
Aurora Cannabis Announces Financial Results for the Third Quarter of Fiscal 2019
- Solid Net Revenue Growth Across All Channels to $65.1 Million
- Production Volumes Double While Per-Unit Production Costs Decline
- On Track to Deliver Positive EBITDA Beginning in Fiscal Q4 2019
EDMONTON, May 14, 2019 /PRNewswire/ – Aurora Cannabis Inc. (the “Company” or “Aurora”) (NYSE:ACB) (TSX: ACB), announced today its financial and operational results for the third quarter ended March 31st, 2019.
Third Quarter 2019 Highlights
Unless otherwise stated, comparisons are made between Fiscal Q3 2019 and Fiscal Q2 2019 results)
- Continued solid revenue growth averaging 20% across all key markets, driven by successful scale up of the Company’s production and continued strong performance across the Canadian consumer, and Canadian and International medical cannabis markets:
- Canadian Consumer up 37%
- Canadian Medical up 8%
- International Medical up 40%
- Growth of the Company’s medical patient base, up by 5% to 77,136. As at the date of this release, Aurora has 82,745 active registered patients, a further increase of 7%, and continues to register new patients as product availability ramps up.
- Cash cost to produce per gram declined 26% to $1.42 per gram, as the initial impact of Aurora Sky’s scale and efficiency began to be realized.
- Production volume increased 99% to 15,590 kgs, up 1,200% year-over-year. The increase in production accelerated through the quarter, with the majority of the harvested volume realized in the last half of the quarter.
- SG&A expenses have stabilized with a modest increase of 1%, reflecting Aurora’s ongoing commitment to disciplined cost management.
- Average selling price per gram decreased marginally due to product mix effects (higher contribution from wholesale consumer), extraction capacity constraints resulting in extract-based products comprising 18% of net cannabis sales, and the first full quarter impact of excise tax on medical cannabis.
- Adjusted EBITDA loss improved by 20% to $36.6 million as the company continues to track towards achieving EBITDA positive results beginning in Q4 2019 as operations continue to ramp up.
- In January 2019, Aurora completed a US$345 million Convertible Notes offering, with the proceeds earmarked to continue the Company’s pace of growth in Canada and internationally. IFRS accounting standards require a mark-to-market adjustment at each period end for the derivative portion of these notes. Due to the increase in Aurora’s stock price since the issuance of the notes, the Company recorded a $102 million non-cash fair value loss in the Q3 2019 profit and loss statement.
Management Commentary
I’m exceptionally proud of our company and team as Aurora continues to deliver on our domestic and international growth strategy. We achieved solid revenue growth and strong operating results in a quarter proven challenging across the industry. We are laser focused on building a long-term sustainable business.
 Terry Booth, CEO
Terry Booth, CEO
During the quarter, we formally welcomed Nelson Peltz a key strategic advisor. He has been incredibly engaged, collaborative, and strategically focused on assisting our pursuit of growth in global markets and with mature companies in adjacent industries.
Glen Ibbott, CFO, added, “Aurora is an extremely active and diversified company, leading the industry in cannabis research, product development, cultivation, global scale, and revenue growth. With a solid Q3 on all fronts, it’s time to move the yardsticks for the industry again. The company we have built with purpose through both organic growth and targeted acquisitions has provided a unique opportunity: continue to lead the industry in revenue growth while also progressing to positive operating earnings in the near term.”
Q3 2019 Key Financial and Operational Metrics
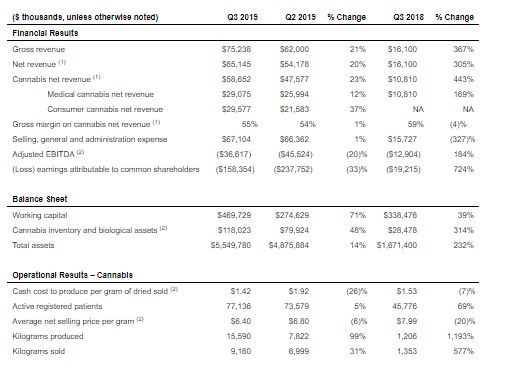
¹Net revenue represents our total gross revenue exclusive of excise taxes levied by the Canada Revenue Agency (“CRA”) on the sale of medical and recreational cannabis products effective October 17, 2018.
²These terms and non-GAAP measures are defined or reconciled in Aurora’s Q3 2019 MD&A.
Outlook
The Aurora Sky and Bradford facilities are now operating at full capacity. With this, the Company’s annualized production run rate across its operational facilities is in excess of 150,000 kg per annum, based on planted rooms.
Aurora reiterates its target for Q4 with production available for sale in excess of 25,000 kg. Management intends to allocate a portion of this capacity to its inventory for manufacturing new products. Aurora remains focused on having vapes and certain edibles ready for launch under new regulations in the Canadian consumer market which are expected toward the end of the calendar year.
With production ramping up, the Company continues to scale up manufacturing capacity, with innovation and technologies aimed at reducing time from harvest to market. The Company anticipates that increased processing, packaging and delivery efficiencies in Q4 and beyond will accelerate availability of product.
Supply to Europe and other international markets is expected to increase as more of Aurora’s production facilities receive EU GMP certification. The Bradford facility has recently undergone an audit to obtain EU GMP certification. In Q3, the Company began exports of full spectrum cannabis extracts in Germany. Management anticipates these sales will contribute to growth given the higher margins in extracts.
Oil extraction capacity has been a constraint during the second and third fiscal quarters of 2019. Subsequent to quarter’s end, Aurora expanded its internal extraction capacity to almost 7,000 kgs per quarter currently and will reach almost 16,000 kgs per quarter in fiscal Q1. As well, the Company’s extraction partner Radient Technologies is scaling up commercial production at its Edmonton facility. Consequently, Aurora anticipates production of extract-based products to increase, with the full impact starting to materialize towards the end of fiscal Q4. This increase in internal and external extraction capacity will enhance Aurora’s ability to produce derivative products at scale, which management expects will have a positive impact on both revenues and gross margin.
With Aurora Sky now operating at full capacity, the Company anticipates continued reduction in production and manufacturing costs allowing cash costs per gram to continue to trend lower. Management reiterates its expectation that the average cash cost to produce per gram at its Sky Class facilities will be below $1.
With disciplined cost management, the Company expects SG&A costs to grow modestly over the remainder of the fiscal year. Consequently, management anticipates that with sustained revenue growth and lower cash costs per gram, Aurora is well positioned to achieve positive EBITDA beginning in fiscal Q4 2019 (calendar Q2 2019).
Q3 2019 Facility and Production Update
Aurora defines production rate as the capacity of all planted rooms that have been approved by the regulator for sales, using anticipated annualized harvests at maturity based on a historical yield per plant. These targeted yields have been met or exceeded at all of Aurora’s current operating facilities. To view a video overview of Aurora’s production facilities:
- Construction at Aurora Sky is complete, and all grow rooms have been licensed by Health Canada. Fully planted, Aurora Sky is operating at its full design capacity of over 100,000 kg per annum.
- All rooms at Bradford are licensed by Health Canada and the facility has been fully planted. The facility has received no major observations during its audit to obtain EU GMP certification. Obtaining this certificate will substantially increase the Company’s capacity to ship product to the European market.
- The first saleable harvest at Aurora Nordic 1 is expected by the end of fiscal Q4 2019, with product sales anticipated in December 2019, or as soon as regulatory approvals are provided. Initial harvests for testing and licensing purposes have been completed successfully.
- Construction of Aurora Sun is progressing well, with the facility anticipated to be ready for planting by mid calendar 2020. Aurora Sun will measure 1.62 million square feet, reflecting a 33% increase from its originally planned size.
- Erection of the steel structure is well underway and anticipated for completion in late June 2019.
- Construction of the glass roof has commenced and is anticipated for completion shortly after installation of the steel structure.
- Whistler Alpha Lake is currently operating at its designed capacity of 480 kg/year of organic certified cannabis, utilizing four grow rooms.
- Construction of Whistler Pemberton remains on track for completion in calendar Q4 2019
- Four rooms are fully operational with an annual capacity of 1,200kg of organic certified cannabis
- 11 additional rooms are expected to come online beginning in November 2019
- Upon completion Pemberton is expected to produce 4,500 kg/year of organic certified cannabis
- The Pemberton facility will incorporate a public lounge to educate visitors about WMMC and its history as cultivation pioneers of organic certified cannabis
- During the most recent grow season in Europe, Agropro harvested a combined 3,950 acres of hemp across Lithuania, Latvia and Estonia. For the upcoming grow season, beginning in May 2019, Agropro plans to contract 8,150 acres of hemp for harvest, which is expected to begin in August.
Q3 2019 and Subsequent Corporate Highlights
Acquisitions
- Acquisition of Whistler Medical Marijuana Corporation (“Whistler”)
On March 1, 2019, Aurora acquired Whistler, an iconic Canadian organic cannabis brand which commands a significant premium for its products in both the Canadian medical and consumer markets. The Company is currently scaling up Whistler’s operations and anticipates an increase in products available for sale for the remainder of the calendar year.
- Acquisition of Hempco Food and Fiber Inc. (“Hempco”)
On April 16, 2019, the Company entered a binding letter agreement with Hempco to acquire all of the issued and outstanding shares of Hempco. The acquisition will strengthen the Company’s industrial hemp and CBD-from-hemp infrastructure.
- Acquisition of Chemi Pharmaceuticals Inc. (“Chemi”)
On April 24, 2019, the Company acquired Chemi, an Ontario-based laboratory specializing in high quality analytics services for the pharmaceutical and cannabis industries. The acquisition is intended to expand the Company’s analytical services for derivative products.
Strategic Developments
- Appointment of Nelson Peltz
On March 13, 2019, the Company appointed Nelson Peltz as a strategic advisor to work collaboratively with Aurora on exploring global expansion and partnership opportunities. Management believes the Company is well positioned to pursue partnerships across a number of industry verticals and is working with Mr. Peltz and his team to assess opportunities.
- Aurora Polaris
On February 12, 2019, the Company announced the construction of Aurora Polaris, a 300,000 square foot international logistics hub and facility for the industrial-scale production of derivative cannabis products. Construction is on schedule and the Company anticipates completion towards the end of the calendar year. In anticipation of new regulations, the Company is in the process of installing interim production lines in licensed space across its production infrastructure in Canada to ensure a full complement of products will be available for sale in substantial quantities when permitted.
International Expansion
- German Cannabis Production Tender
On April 5, 2019, the Company was selected by the German Federal Institute for Drugs and Medical Devices as one of three winners in a public tender to cultivate and distribute medical cannabis in Germany. Aurora scored highest across 11 of 13 tender lots and was allotted the maximum number given to any LP of five. Aurora has commenced work on the construction of a facility in Germany and anticipated product to reach the German market in October 2020. Management expects that becoming a local producer will strengthen brand awareness and market development in a large and important market.
- Exports of Medical Cannabis to the United Kingdom
On February 11, 2019, the Company completed its first commercial export of cannabis oil to the United Kingdom. Under the new UK framework, specialist doctors can legally issue prescriptions for cannabis-based medicines when they agree that their patients could benefit from this treatment.
- Expansion into Portugal
On February 26, 2019, the Company created Aurora Portugal Lda. through an agreement to acquire a 51% ownership interest in Gaia Pharm Lda. Construction of an EU GMP compliant production cannabis facility is now underway in Portugal.
Exports of Cannabis Oil to Germany
On March 11, 2019, the Company commenced sales of cannabis oils to German pharmacies. Aurora’s full spectrum extract is differentiated in a market predominantly serviced with synthetic cannabinoids. With this, management believes the Company has a significant competitive advantage to establish early mover leadership, brand awareness and sales growth in this higher margin segment.
Financing Activities
- Offering of Convertible Notes
On January 24, 2019, the Company closed an offering of Convertible Notes for gross proceeds of US$345 million to fuel Canadian and international expansion initiatives, for future acquisitions and for general corporate purposes, including working capital requirements to continue the Company’s accelerated growth.
Filing of Final Base Shelf Prospectus and Prospectus Supplement for At-the-Market Offering
On May 10, 2019, the Company filed a final short-form base shelf prospectus (the “Shelf Prospectus”) with the securities commissions in each of the provinces of Canada, except Quebec, and a corresponding shelf registration statement with the United States Securities and Exchange Commission on Form F-10. These filings allow the Company to qualify the distribution under a prospectus in Canada and the United States of up to US$750,000,000 of common shares, warrants, subscription receipts, debt securities, or any combination of such securities (all of the foregoing, collectively, the “Shelf Securities”) during the 25-month period that the final short form base shelf prospectus remains effective. The specific terms of any future offerings under the Shelf Prospectus will be established in a prospectus supplement. Any prospectus supplement will be filed with the applicable securities regulatory authorities in connection with such offering.
- Filing of Final Base Shelf Prospectus and Prospectus Supplement for At-the-Market Offering
On May 10, 2019, the Company filed a final short-form base shelf prospectus (the “Shelf Prospectus”) with the securities commissions in each of the provinces of Canada, except Quebec, and a corresponding shelf registration statement with the United States Securities and Exchange Commission on Form F-10. These filings allow the Company to qualify the distribution under a prospectus in Canada and the United States of up to US$750,000,000 of common shares, warrants, subscription receipts, debt securities, or any combination of such securities (all of the foregoing, collectively, the “Shelf Securities”) during the 25-month period that the final short form base shelf prospectus remains effective. The specific terms of any future offerings under the Shelf Prospectus will be established in a prospectus supplement. Any prospectus supplement will be filed with the applicable securities regulatory authorities in connection with such offering.
On May 14, 2019, the Company filed a prospectus supplement (the “Prospectus Supplement”) to the Shelf Prospectus.In connection with the Prospectus Supplement, Aurora entered into a Sales Agreement dated May 14, 2019 with Cowen and Company, LLC (“Cowen”) and BMO Capital Markets (“BMO”) who will act as the selling agents (the “Selling Agents”) for the sale of common shares of Aurora (the “Common Shares”) by way of “at-the-market distributions” on the New York Stock Exchange in the United States. Subject to the terms of the Sales Agreement and applicable regulatory requirements, Common Shares in the aggregate amount of up to US$400,000,000 may be issued and sold from time to time at the discretion of Aurora over a period of up to 25 months. The Common Shares will be distributed at market prices prevailing at the time of the sale of such Common Shares and, as a result, prices may vary as between purchasers and during the period of distribution. The net proceeds of such sales, if any, will be used for general corporate purposes, including: (i) working capital; (ii) potential future acquisitions; (iii) debt repayments; and (iv) capital expenditures. The volume and timing of sales, if any, of Common Shares is at the discretion of Aurora.
Aurora expects to use the net proceeds from the prospectus will support its expansion initiatives, global partnership strategy, and to continue the Company’s accelerated growth.
The Shelf Prospectus and Prospectus Supplement have been filed on SEDAR and the U.S. version of the Shelf Prospectus and the Prospectus Supplement have been filed on the SEC’s website (www.sec.gov).
This news release does not constitute an offer to sell or the solicitation of an offer to buy, nor shall there be any sale of these Securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to the registration or qualification under the securities laws of any such jurisdiction.
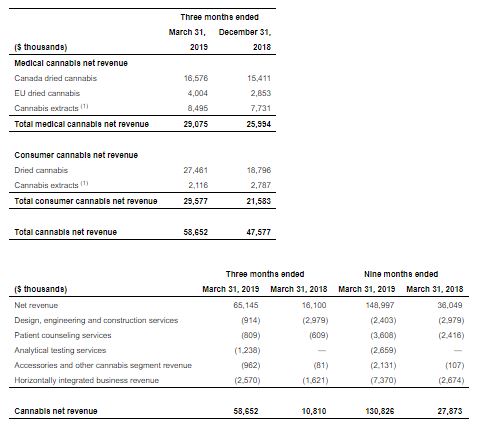
Financial Supplement Q3 2019
Options Grant
The Company granted a total of 383,000 options to purchase common shares of Aurora to Directors and Officers of the Company. The options vest annually over 36 months and have a weighted average exercise price of $9.35 per common share.
Conference Call
Aurora will host a conference call tomorrow, May 15, 2019, to discuss these results. Terry Booth, Chief Executive Officer, Glen Ibbott, Chief Financial Officer, Cam Battley, Chief Corporate Officer, and Michael Singer, Executive Chairman, will host the call starting at 10:30 a.m. Eastern time. A question and answer session will follow management’s presentation.
Date: Wednesday, May 15th, 2019
Time: 10:30 a.m. Eastern Time | 8:30 a.m. Mountain Time
Webcast: https://bit.ly/2GRpDP3
Replay: (416) 849-0833 or (855) 859-2056 until 12:00 midnight Eastern Time Wednesday, May 22, 2019
Reference Number: 1103129
¹Non-IFRS measures are defined in the company’s MD&A.
About Aurora
Headquartered in Edmonton, Alberta, Canada with funded capacity in excess of 625,000 kg per annum and sales and operations in 24 countries across five continents, Aurora is one of the world’s largest and leading cannabis companies. Aurora is vertically integrated and horizontally diversified across every key segment of the value chain, from facility engineering and design to cannabis breeding and genetics research, cannabis and hemp production, derivatives, high value-add product development, home cultivation, wholesale and retail distribution.
Highly differentiated from its peers, Aurora has established a uniquely advanced, consistent and efficient production strategy, based on purpose-built facilities that integrate leading-edge technologies across all processes, defined by extensive automation and customization, resulting in the massive scale production of high-quality product at low cost. Intended to be replicable and scalable globally, our production facilities are designed to produce cannabis of significant scale, with high quality, industry-leading yields, and low per gram production costs. Each of Aurora’s facilities is built to meet EU GMP standards. EU GMP certification has been granted to Aurora’s first production facility in Mountain View County, the MedReleaf Markham facility, and its wholly owned European medical cannabis distributor Aurora Deutschland.
In addition to the Company’s rapid organic growth and strong execution on strategic M&A, which to date includes 16 wholly owned subsidiary companies – MedReleaf, CanvasRX, Peloton Pharmaceutical, Aurora Deutschland, H2 Biopharma, Urban Cultivator, BC Northern Lights, Larssen Greenhouses, CanniMed Therapeutics, Anandia, HotHouse Consulting, MED Colombia, Agropro, Borela, ICC Labs, Whistler, and Chemi Pharmaceutical – Aurora is distinguished by its reputation as a partner and employer of choice in the global cannabis sector, having invested in and established strategic partnerships with a range of leading innovators, including: Radient Technologies Inc. (TSXV: RTI), Hempco Food and Fiber Inc. (TSXV: HEMP), Cann Group Ltd. (ASX: CAN), Micron Waste Technologies Inc. (CSE: MWM), Choom Holdings Inc. (CSE: CHOO), Capcium Inc. (private), Evio Beauty Group (private), Wagner Dimas (private), CTT Pharmaceuticals (OTCC: CTTH), Alcanna Inc. (TSX: CLIQ), High Tide Inc. (CSE: HITI) and EnWave Corporate (TSXV: ENW).
Aurora’s Common Shares trade on the TSX and NYSE under the symbol “ACB”, and are a constituent of the S&P/TSX Composite Index.
For more information about Aurora, please visit our investor website, investor.auroramj.com
