 MedReleaf Reports Third Quarter Fiscal 2018 Results
MedReleaf Reports Third Quarter Fiscal 2018 Results
Extract growth drives record sales and volume sold
MARKHAM, ON, Feb. 13, 2018 /CNW/ – MedReleaf Corp. (TSX: LEAF) (“MedReleaf” or the “Company”), Canada’s first and only ISO 9001 and ICH-GMP certified cannabis producer, today announced financial and operating results for the third quarter fiscal 2018 ending December 31, 2017. All amounts expressed are in Canadian dollars unless otherwise noted.
In the third quarter, we set new records for total sales and grams sold demonstrating the strength of our underlying business while we continue to make investments in preparation for the recreational market and for international expansion. These investments are starting to bear fruit and we now have boots on the ground in six different countries working on various initiatives – both for cultivation and export.
 Neil Closner, CEO of MedReleaf.
Neil Closner, CEO of MedReleaf.
We also launched our first adult recreational-use brand, San Rafael ’71, which is getting good reception from the various Provincial purchase authorities. With a strong balance sheet bolstered by $192.5 million in equity capital raised since last December, we have a clear opportunity to establish MedReleaf as a global leader in the cannabis industry.
Third Quarter Fiscal 2018 Financial Summary
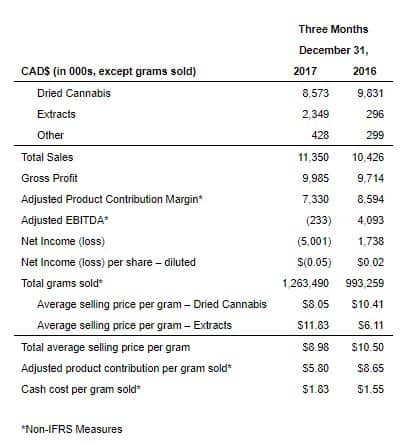
Third Quarter Fiscal 2018 Financial Highlights
- Record sales of $11.4 million, an increase of 9% from the prior year period, and a 16% increase from the second quarter of fiscal 2018.
- Sold a record 1,263 kilograms of cannabis products, an increase of 27% from the prior year period and a 20% increase from the second quarter of fiscal 2018.
- Sales of cannabis-based extract products were $2.3 million, or 21% of total sales.
- Adjusted EBITDA loss of $0.2 million, a decrease of $4.3 million from the prior year period due to discounts offered to qualifying Veterans as a result of the Veterans Affairs Canada (VAC) reimbursement policy change, overhead costs associated with the Bradford Facility construction, and operating investments towards future growth.
- Average selling price per gram of $8.98, a decrease from $10.50 for the prior year period due to the reduction in VAC reimbursement pricing.
- Cash cost per gram sold of $1.83, an increase from $1.55 for the prior year period primarily due to increased plant operating costs and fixed overhead attributable to the Bradford Facility. Cash cost per total gram sold is expected to improve as greater efficiencies of scale are realized when construction of the Bradford Facility is fully completed.
- Adjusted product contribution margin per gram sold of $5.80, a decrease from $8.65 for the prior year period.
Third Quarter Fiscal 2018 Business Highlights
- On November 14, 2017, MedReleaf’s Australian join venture partner, Indica Industries Pty Ltd. (t/a “MedReleaf Australia”), was granted a licence from the Australian Government Office of Drug Control (“ODC”) for the cultivation and production of medical cannabis.
- On November 30, 2017, MedReleaf won 10 awards at the 2017 Lift Canadian Cannabis Awards including Top Licensed Producer and first place product awards for: Top Indica Flower, Top High CBD Flower, Top High THC Flower, and Top High CBD Oil.
- On December 4, 2017, the Company closed a bought deal equity financing for gross proceeds of approximately $60.0 million.
- On December 21, 2017, the Company announced an agreement to become a medical cannabis supplier to Shoppers Drug Mart.
Subsequent to the Third Quarter Fiscal 2018
- As at February 2018, the total annual production capacity at the Company’s Bradford Facility is 9,500 kilograms, including 3,900 kilograms of capacity added since November 2017 that is pending license approval from Health Canada. The Company remains on schedule to complete construction of the Bradford Facility with annual capacity totalling 28,000 kilograms by the summer of 2018.
- On January 31, 2018, the Company closed a bought deal equity financing for gross proceeds of approximately $132.5 million.
- On February 6, 2018, MedReleaf announced it has received Health Canada approval for the sale of its cannabis oil softgel capsules, becoming the first LP to bring colour-coded and cannabis variety-specific softgel capsules to market.
- On February 8, 2018, MedReleaf introduced its first adult-use recreational brand, San Rafael ’71, inspired by and designed to celebrate the spirit of cannabis culture with the classic consumer in mind – one the largest segments of the Canadian cannabis market.
Financial Review
Sales
Sales for the third quarter of fiscal 2018 reached a new all-time high of $11.4 million, an increase of 9% from the prior year period, and a 16% increase from the second quarter of fiscal 2018. The increase in sales from the prior year period was primarily due to sales growth for extract-based products partially offset by Veteran volume capacity and pricing limitations that impacted sales of dried cannabis products. Sales of dried cannabis for the third quarter of fiscal 2018 were $8.6 million, a decrease of 13% from the prior year period.
Extract sales for the third quarter were $2.3 million, an increase of $2.1 million from the prior year period, and represented 21% of total sales. With the launch of topical creams, softgel capsules, future product development initiatives, and growing industry demand, MedReleaf expects sales of extract products to account for an increasing percentage of the Company’s overall revenue in the future.
During the third quarter of fiscal 2018, a total of 1,263.5 kilograms of cannabis products were sold, an increase of 27% from the prior year period, and an increase of 20% from the second quarter of fiscal 2018.
Total average selling price for the third quarter of fiscal 2018 was $8.98 per gram compared to $10.50 for the prior year period. The reduction in average selling price per gram from the prior year period is a result of discounts offered to qualifying Veterans due to the VAC Policy change that provides for a maximum reimbursement rate of $8.50 per gram effective November 22, 2016.
Cash Cost Per Gram Sold (Non-IFRS Measure)
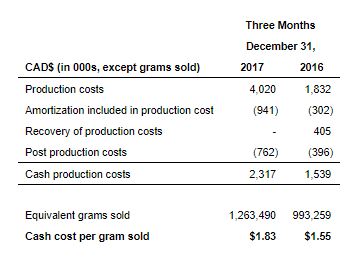
The following are the Company’s cash production costs, on a total and per gram sold basis, for the three and months ended December 31, 2017 and 2016, as compared to reported production costs (excluding costs resulting from the fair value of biological assets), which represents cost of sales, in accordance with IFRS:
The cash cost per gram sold for the third quarter of fiscal 2018 was $1.83 compared to cash cost per gram sold of $1.55for the prior year period. The increase in cash cost per gram is primarily due to increased plant operating costs and fixed overhead attributable to the Bradford Facility. Cash cost per total gram sold is expected to decrease steadily as increased production and yield improvements are realized from the completed construction of additional cultivation capacity at the Bradford Facility.
Adjusted Product Contribution Margin (Non-IFRS Measure)
The following is the Company’s Adjusted Product Contribution Margin as compared to the reported gross profit, which includes the gain on changes in fair value of biological assets in accordance with IFRS, for the three months ended December 31, 2017 and 2016.
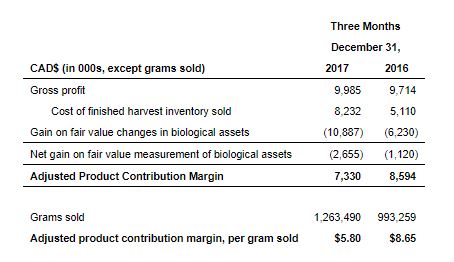
Adjusted Product Contribution Margin for the third quarter of fiscal 2018 was $7.3 million or $5.50 per gram sold, compared to $8.6 million or $8.65 per gram sold for the prior year period.
The decrease in Adjusted Product Contribution Margin and Adjusted Product Contribution Margin per gram sold from the prior year period was the result of increased labour costs and depreciation attributable to the Bradford Facility expansion in addition to both price and volume limits imposed by VAC Policy change.
Adjusted EBITDA (Non-IFRS Measure)
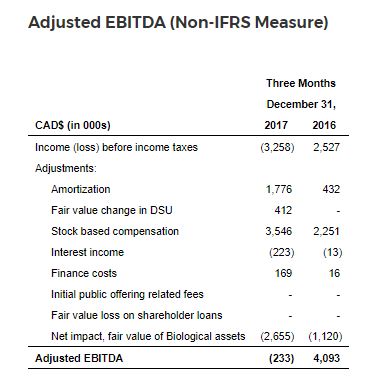
Adjusted EBITDA for the third quarter of fiscal 2018 was ($0.2) million, a decrease of $4.3 million from $4.1 million for the prior year period.
The decrease in Adjusted EBITDA for the third quarter of fiscal 2018 compared to the prior year period is the result of: overhead costs incurred to support the Bradford Facility; increased expenditures related to professional fees; business development; increased patient support costs to support patient demand; investments in sales, marketing and brand development; increased human resource talent to support current and future growth; and as a result of the VAC Policy, the Company offering discounts to qualifying Veterans to assist with the non-reimbursable portion of their medication.
Net Income
Net loss for the third quarter of fiscal 2018 was $5.0 million compared to net income of $1.7 million for the prior year period. The net income decrease was mainly due to costs incurred related to the Company’s stock based compensation expense, fair value loss related to the Company’s deferred share unit (DSU) plan, increased operating and overhead expenses, and increased advertising and promotional expenses.
Balance Sheet
At the end of December 31, 2017, the Company had cash and cash equivalents of $114.6 million, working capital of $137.9 million.
Inventories as at December 31, 2017 were $24.9 million, an increase of $15.4 million from March 31, 2017 due primarily to inventory produced at the Bradford Facility. Of the total increase in inventories, $10.7 million was due to changes in the fair value associated with the deemed cost of inventory, the balance was due primarily to the production of work-in-process dried cannabis leaf product produced for future extraction. As at December 31, 2017, the equivalent of approximately seven months of cost of sales are included in inventory.
Biological assets as at December 31, 2017 were $3.8 million, an increase of $1.0 million compared to March 31, 2017 of $2.8 million. This increase was due to increased fair value gains on biological assets resulting from increased yields and the addition of biological assets at the Bradford Facility.
Cash flow used in operating activities for the nine months ended December 31, 2017 was $0.8 million compared cash flow provided by operating activities of $15.8 million for the prior year period. The decrease in cash flow provided by operating activities was mainly due to IPO related costs and increased operating costs, that were partially offset by increased sales and gross profit. Increase in sales was due primarily to patient demand, yield improvements, and the introduction of extracted oil sales in November of 2016.
Capital expenditures for the nine months ended December 31, 2017 were $29.8 million primarily put towards production rooms, building improvements, furniture and other equipment related to the construction and development of the Bradford facility. As at December 31, 2017, approximately $9.0 million of future payments have been committed in relation to the development of the Bradford Facility.
MedReleaf is fully funded for its announced plans to increase capacity to 35,000 kilograms in production annually. The Company has a fully drawn $10 million term credit facility and an undrawn $10 million revolving credit facility with CIBC. In addition, MedReleaf completed two bought deal equity financings, one on December 4, 2017 and a second on January 31, 2018, for total gross proceeds of approximately $192.5 million. The proceeds from these two equity financings will be used to finance the acquisition and/or construction of additional cannabis production and manufacturing facilities and expand the Company’s marketing and sales initiatives.
Third Quarter Fiscal Year 2018 Conference Call & Webcast
A conference call and webcast to discuss MedReleaf’s second quarter fiscal year 2018 results will be held on Tuesday, February 13, 2018 at 8:00 a.m. (ET). The call will be hosted by Neil Closner, Chief Executive Officer, and Igor Gimelshtein, Chief Financial Officer, followed by a question and answer period.
To participate, interested parties are asked to dial (647) 427-7450 or (888) 231-8191 prior to the scheduled start of the call. A replay of the conference call will be available by dialing (855) 859-2056 and using the reference number 2397185. The replay of this call will be available until February 20, 2018.
The Conference Call will also be webcast live at http://bit.ly/2DQpYSM
Financial Statements and Management’s Discussion and Analysis
This news release, along with the unaudited condensed interim consolidated financial statements for the three and nine month periods ended December 31, 2017 and 2016, including the notes thereto, and the Company’s corresponding management’s discussion and analysis, are available on the Company’s website at www.medreleaf.com and on SEDAR at www.sedar.com.
Non-IFRS Measures
This news release refers to certain non-IFRS financial measures. These measures are not recognized measures under IFRS, do not have a standardized meaning prescribed by IFRS and are therefore unlikely to be comparable to similar measures presented by other companies. Rather, these measures are provided as additional information to complement those IFRS measures by providing additional information regarding the Company’s results of operations from management’s perspective. Accordingly, non-IFRS measures should not be considered in isolation nor as a substitute for analysis of the Company’s financial information reported under IFRS. All non-IFRS measures presented in this news release are reconciled to their closest reported IFRS measure.
(a) Adjusted Product Contribution Margin
Management makes use of an “Adjusted Product Contribution Margin” measure to provide a better representation of performance in the period by excluding non-cash fair value measurements as required by IFRS. Management believes this measure provides useful information as it represents the gross margin for management purposes based on the Company’s complete cost to produce inventory sold, exclusive of any fair value measurements as required by IFRS. The metric is calculated by removing all amounts related to biological asset fair value accounting under IFRS including gains on transformation of biological assets and the cost of finished harvest inventory sold, which represents the fair value measured portion of inventory cost (“fair value cost adjustment”) recognized as cost of goods sold.
(b) Equivalent grams and kilograms
Equivalent gram or kilogram refers to the equivalent number of dried grams or kilograms of cannabis required to produce extracted cannabis in the form of cannabis oil. The Company estimates and converts its cannabis oil inventory to equivalent grams using the combined Tetrahydrocannabinol (“THC”) and Cannabidiol (“CBD”) content in extracted cannabis products. Any reference to grams in this news release includes the combined dried cannabis and equivalent grams of extracted cannabis.
(c) Cash Cost Per Gram Sold
The cash cost per gram sold is used by management to measure the estimated amount of direct production costs, on a per gram sold basis, that are required to produce dried cannabis and cannabis oil. Management uses this measure to track production cost trends and assess the sensitivity and tolerance for pricing changes. Management believes this measure provides useful information by removing non-cash and post production costs and provides a benchmark of the Company against its competitors. The metric is calculated by: removing from production costs incurred during the period, all non-cash based costs (including amortization and inventory write-downs or impairments) and all post production costs; and dividing such amount by the approximate number of grams of cannabis sold during the period. Post production costs include indirect overhead expenses such as: equipment rentals, payment processing fees, indirect labour expenses, shipping expenses, quality control, expenses, and other order fulfillment costs included in production costs.
(d) Adjusted Earnings Before Interest, Tax, Depreciation and Amortization (“Adjusted EBITDA”)
Adjusted EBITDA is used by management as a supplemental measure to review and assess operating performance and trends on a comparable basis. The Company defines Adjusted EBITDA as EBITDA adjusted for the impact of any unrealized expenses or gains, stock based compensation, fair value gains or costs arising from biological assets, expenses related to readying the Company for its initial public offering and other non-recurring costs the Company deems unrelated to current operations.
The Company believes that Adjusted EBITDA provides a useful tool for assessing the comparability between periods of its ability to generate cash from operations. Adjusted EBITDA is presented in order to provide supplemental information to the Financial Statements included elsewhere in this MD&A, and such information is not meant to replace or supersede IFRS measures.
About MedReleaf Corp.
Voted Top Licensed Producer at the 2017 Lift Canadian Cannabis Awards, MedReleaf is an R&D-driven company dedicated to innovation, operational excellence and the production of top-quality cannabis. Sourced from around the world and carefully cultivated in one of two state of the art ICH-GMP and ISO 90001 certified facilities in Ontario, the Company delivers a variety of premium products for the global medical market and is committed to serving the therapeutic needs of its medical patients and providing a compelling product assortment for the adult-use recreational consumer.
For more information on MedReleaf, its products, research and how the company is helping patients #livefree, please visit MedReleaf.com or follow @medreleaf
