GW Pharmaceuticals plc Reports Financial Results and Operational Progress for the Third Quarter Ended September 30, 2019
- Epidiolex U.S. year to date net sales of $188.0 million, including Q3 net sales of $86.1 million
- Conference call today at 4:30 p.m. ET
CARLSBAD, Calif., Nov. 05, 2019 (GLOBE NEWSWIRE) — GW Pharmaceuticals plc (NASDAQ: GWPH, GW, the Company or the Group), the world leader in the discovery, development and commercialization of cannabinoid prescription medicines, announces financial results for the third quarter ended September 30, 2019.
In this first year of launch, we are pleased to report continued Epidiolex revenue growth in the US. Receptivity to the introduction of this breakthrough treatment continues to be highly encouraging as a result of positive physician and patient experiences as well as strong payer coverage. We see significant opportunity for the short, medium and long term and believe that all the fundamentals are in place to make Epidiolex a very successful brand.
 Justin Gover, GW’s Chief Executive Officer
Justin Gover, GW’s Chief Executive Officer
We can expect to see additional momentum from Europe as well as the launch of the Tuberous Sclerosis indication during 2020. On top of this, GW is ideally placed to consolidate its leadership in cannabinoid science through advancing several mid and late stage pipeline programs in the months ahead.
OPERATIONAL HIGHLIGHTS
- Epidiolex® (cannabidiol)
- U.S. commercial update
- Q3 Net sales of $86.1M; $188.0M net sales in the first three quarters of 2019
- Over 15,000 patients have received Epidiolex prescriptions since launch
- Over 3,000 physicians have generated dispensed prescriptions since launch
- Strong payor coverage with approximately 93 percent of all Commercial, Medicaid and Medicare lives in the US having a coverage determination, of which 65 percent are PA to indication or less restrictive
- European launch update
- European Commission approval in September 2019
- Commercialization underway in France and Germany
- UK NICE guidance expected in Q4 2019; Spain and Italy launches to follow in 2020
- Early Access Program now includes over 1,100 patients across 5 major EU countries, and over 400 physicians from 250 top epilepsy centers
- New Indications
- Tuberous Sclerosis Complex
- TSC Phase 3 data to be presented at American Epilepsy Society annual meeting in December 2019
- TSC sNDA expected to be filed with FDA in early 2020, approval decision expected mid-2020
- TSC EMA submission expected in Q1 2020
- Rett Syndrome
- Phase 3 trial in Rett Syndrome recruiting
- Tuberous Sclerosis Complex
- Life-cycle management
- Several new formulations of CBD in development including modified oral solution, capsule and intravenous formulation
- PK data generated in 2019 supports advancing multiple new formulations into additional Phase 1 studies in 2020
- Several new formulations of CBD in development including modified oral solution, capsule and intravenous formulation
- Exclusivity
- 7 years of orphan exclusivity confirmed by FDA, plus 6-month pediatric extension expected. 10 years of orphan exclusivity in Europe plus 2 year pediatric extension expected
- 9 granted patents listed in “Orange Book” and align directly with Epidiolex FDA label
- Patent expiry dates to 2035
- Additional patent applications under review, including patents related to the use of Epidiolex in TSC and other indications
- Patent application recently published indicating that Epidiolex is more efficacious than synthetic CBD in pre-clinical epilepsy models based on the key difference that Epidiolex comprises up to 2 percent of other cannabinoids.
- Pipeline progress
- Sativex® (nabiximols)
- Initial U.S. target indication: Multiple Sclerosis spasticity
- 3 positive Phase 3 trials completed in Europe
- Clinical IND open, FDA feedback received on clinical plan
- Clinical program expected to commence in Q1 2020 to augment existing pivotal data package
- Clinical program in additional indications in planning for 2020-2021
- Initial U.S. target indication: Multiple Sclerosis spasticity
- CBDV
- IND open for 30-patient open label study in autism expected to commence Q4 19. Initial data in 2020.
- Investigator-led 100 patient placebo-controlled trial in autism underway
- Open label study in Rett syndrome and seizures ongoing
- Neonatal Hypoxic-Ischemic Encephalopathy (NHIE) intravenous CBD program
- Phase 1b safety study in patients expected to commence in Q4
- Orphan Drug and Fast Track Designations granted from FDA and EMA
- Schizophrenia (GWP42003)
- Positive Phase 2a trial published
- Phase 2b trial expected to commence H1 2020
- Sativex® (nabiximols)
- U.S. commercial update
FINANCIAL HIGHLIGHTS
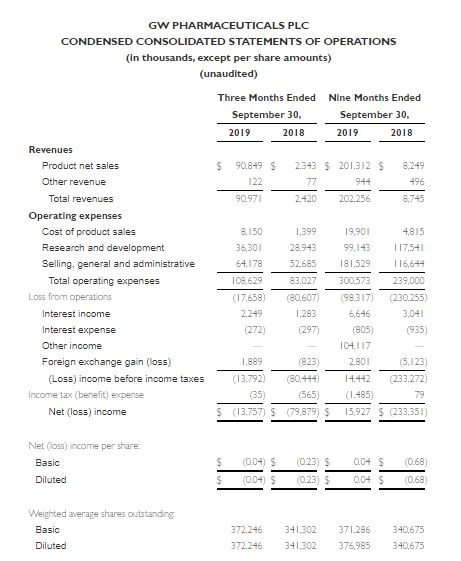
- Revenue for the quarter ended September 30, 2019 was $91.0 million compared to $2.4 million for the quarter ended September 30, 2018
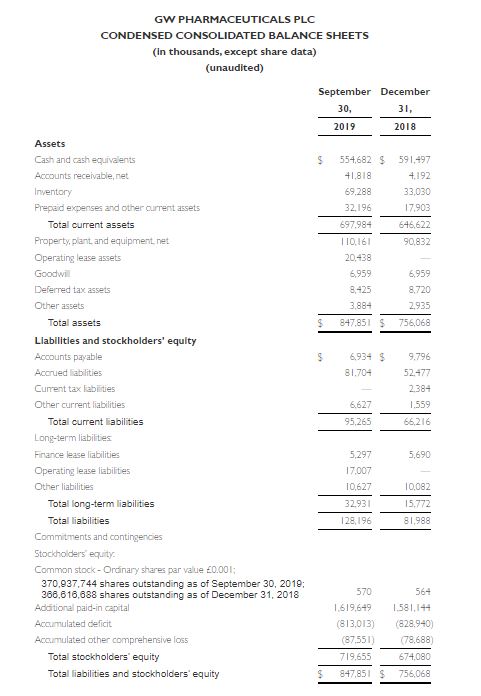
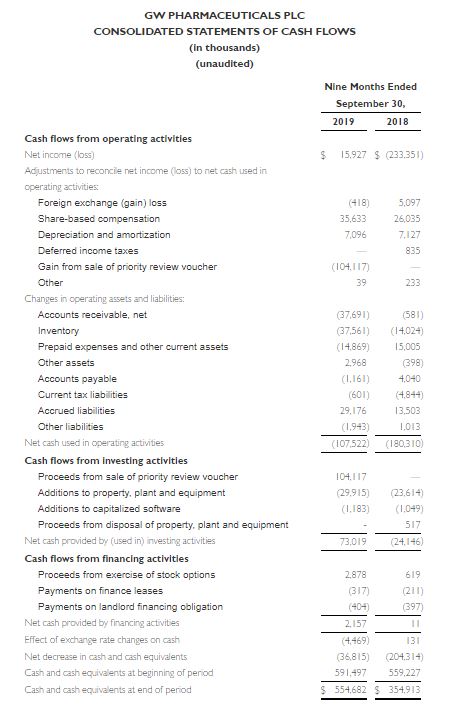
- Cash and cash equivalents at September 30, 2019 were $554.7 million compared to $591.5 million as of December 31, 2018
- Net loss for the quarter ended September 30, 2019 was $13.8 million compared to a net loss of $79.9 million for the quarter ended September 30, 2018
Conference Call and Webcast Information
GW Pharmaceuticals will host a conference call and webcast to discuss the quarter ending September 30, 2019 financial results today at 4:30 pm EST. To participate in the conference call, please dial 877-407-8133 (toll free from the U.S. and Canada) or 201-689-8040 (international). Investors may also access a live audio webcast of the call via the investor relations section of the Company’s website at http://www.gwpharm.com. A replay of the call will also be available through the GW website shortly after the call and will remain available for 90 days. Replay Numbers: (toll free):1-877-481-4010 or 919-882-2331 (international). For both dial-in numbers please use conference Replay ID: 54868.
About GW Pharmaceuticals plc and Greenwich Biosciences, Inc.
Founded in 1998, GW is a biopharmaceutical company focused on discovering, developing and commercializing novel therapeutics from its proprietary cannabinoid product platform in a broad range of disease areas. GW’s lead product, EPIDIOLEX (cannabidiol oral solution) is commercialized in the US by its U.S. subsidiary Greenwich Biosciences for the treatment of seizures associated with Lennox-Gastaut syndrome (LGS) or Dravet syndrome in patients two years of age or older. This product has received approval in Europe under the tradename EPIDYOLEX. The Company continues to evaluate EPIDIOLEX in additional rare conditions including Tuberous Sclerosis Complex (TSC) and Rett syndrome. GW commercialized the world’s first plant-derived cannabinoid prescription drug, Sativex® (nabiximols), which is approved for the treatment of spasticity due to multiple sclerosis in numerous countries outside the United States and for which the Company is now advancing a late stage program in order to seek FDA approval. The Company has a deep pipeline of additional cannabinoid product candidates which includes compounds in Phase 1 and 2 trials for epilepsy, autism, glioblastoma, and schizophrenia. For further information, please visit www.gwpharm.com.