DUBLIN, May 4, 2022 /PRNewswire/ — Jazz Pharmaceuticals plc (Nasdaq: JAZZ) today announced financial results for the first quarter of 2022, raised 2022 financial guidance and provided business updates.
We’re pleased to raise our top- and bottom-line guidance, driven by our continued execution and significant progress across commercial and R&D in the first quarter, which positions us well for the rest of the year and to achieve Vision 2025.
 Bruce Cozadd, chairman and CEO of Jazz Pharmaceuticals
Bruce Cozadd, chairman and CEO of Jazz Pharmaceuticals
Our recent launches of Xywav®, in both narcolepsy and idiopathic hypersomnia (IH), and Rylaze® in acute lymphoblastic leukemia (ALL), continue to generate increased prescriber and patient adoption, and demonstrate our ability to deliver innovative new medicines to improve the lives of patients and their families.
“On the corporate development front, our three recent transactions are aligned with our broader strategy, allowing us to focus on our highest priorities, enhance our pipeline in areas of key interest in neuroscience and oncology and drive long-term shareholder value.”
“We’ve had a highly productive start to 2022 with the submission of two Rylaze Supplemental Biologics License Applications, the first patient enrolled in our Phase 2 basket trial for Zepzelca® and the first presentation of preclinical data for JZP815, an investigational, next-generation pan-RAF kinase inhibitor,” said Rob Iannone, M.D., M.S.C.E., executive vice president, global head of research and development of Jazz Pharmaceuticals. “I’m also excited about the addition of DSP-0187, a potent and highly selective oral orexin-2 receptor agonist, now called JZP441, further strengthening our leadership in sleep medicine, and WTX-613, a differentiated, conditionally activated interferon alpha (IFNα) INDUKINE™ molecule, now called JZP898, which has demonstrated anti-tumor activity in preclinical models.”
These recent transactions reinforce our commitment to enhancing our pipeline and leveraging our productive R&D engine to develop novel medicines for people with serious diseases.
 Rob Iannone
Rob Iannone
- Robust early launch momentum in first full quarter of Xywav for IH
- Submitted a Rylaze Supplemental Biologics License Application (sBLA) for Monday/Wednesday/Friday (M/W/F) intramuscular (IM) dosing and an sBLA for intravenous (IV) administration; both are being reviewed under the Real-Time Oncology Review (RTOR) program
- First patient enrolled in Zepzelca EMERGE-201 Phase 2 basket trial
- Strengthened leadership in sleep medicine with addition of a potent, highly selective oral orexin-2 receptor agonist, JZP441 (DSP-0187)
- Expanded oncology pipeline with JZP898 (WTX-613), a differentiated, conditionally activated IFNα INDUKINE™ molecule
- Strategic divestiture of Sunosi® allows increased investment and sharpens focus on highest strategic priorities
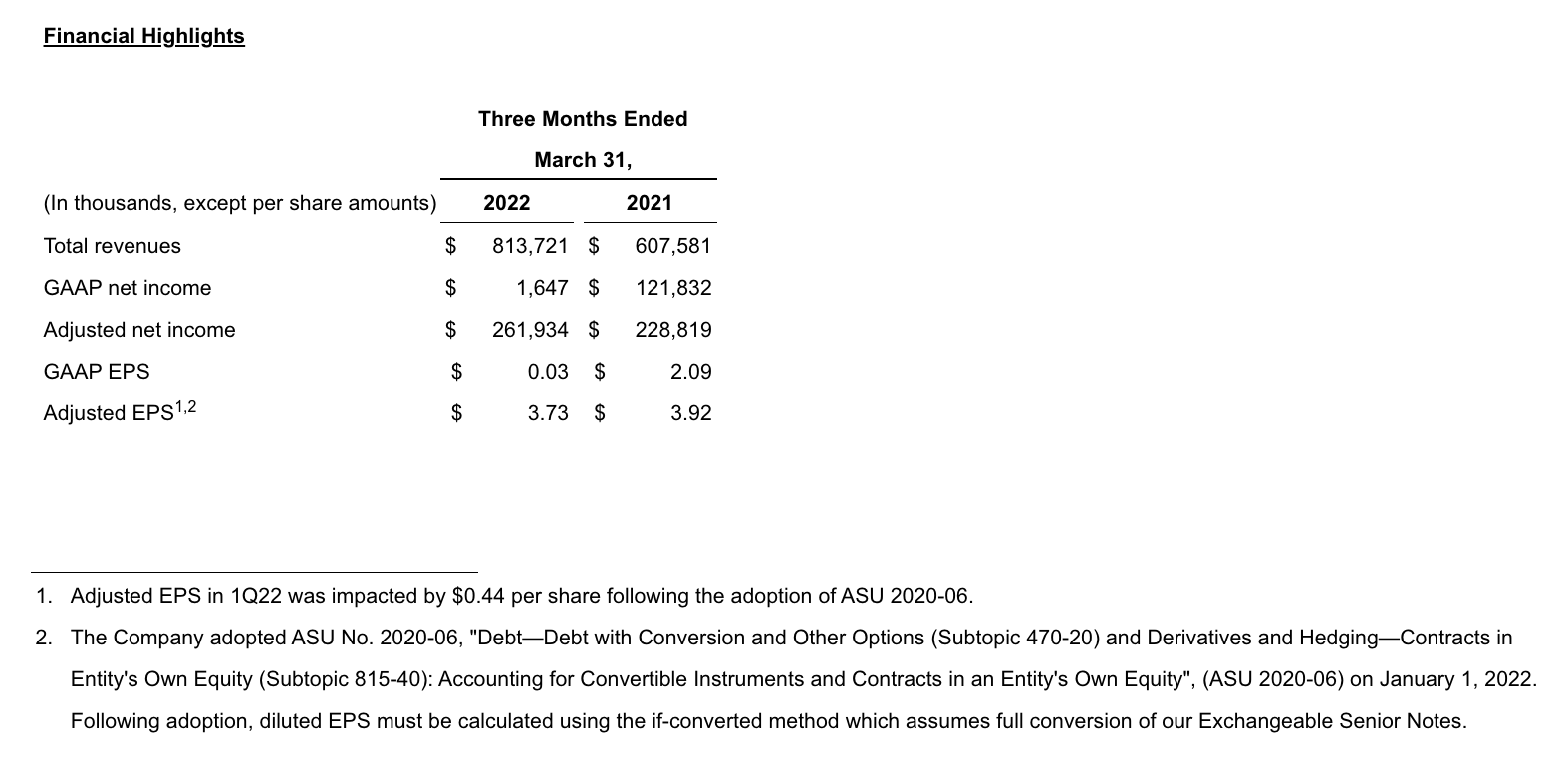
- Growing and durable commercial franchises drove 1Q22 total revenues of $813.7 million; 34% increase compared to the same period in 2021
- Raising top- and bottom-line guidance; 2022 total revenue guidance increased to $3.5 to $3.7 billion
- Net leverage ratio of 3.9x1 as of March 31, 2022, demonstrating rapid deleveraging following the close of the GW Pharmaceuticals (GW) acquisition; on-track for target of less than 3.5x by the end of 2022
- Substantial revenue diversification continues as newer products continue to grow and the Company optimizes its commercial portfolio
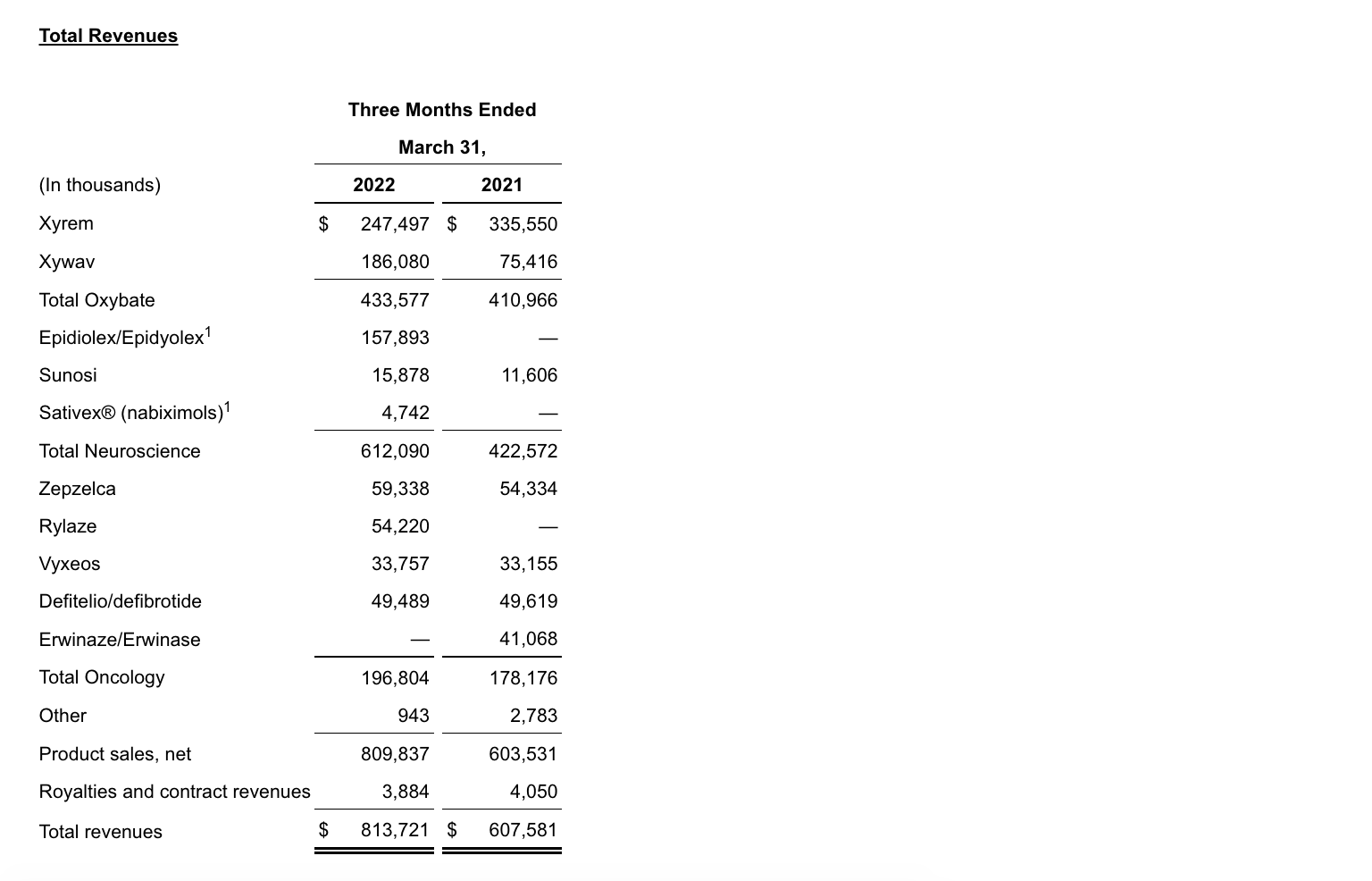
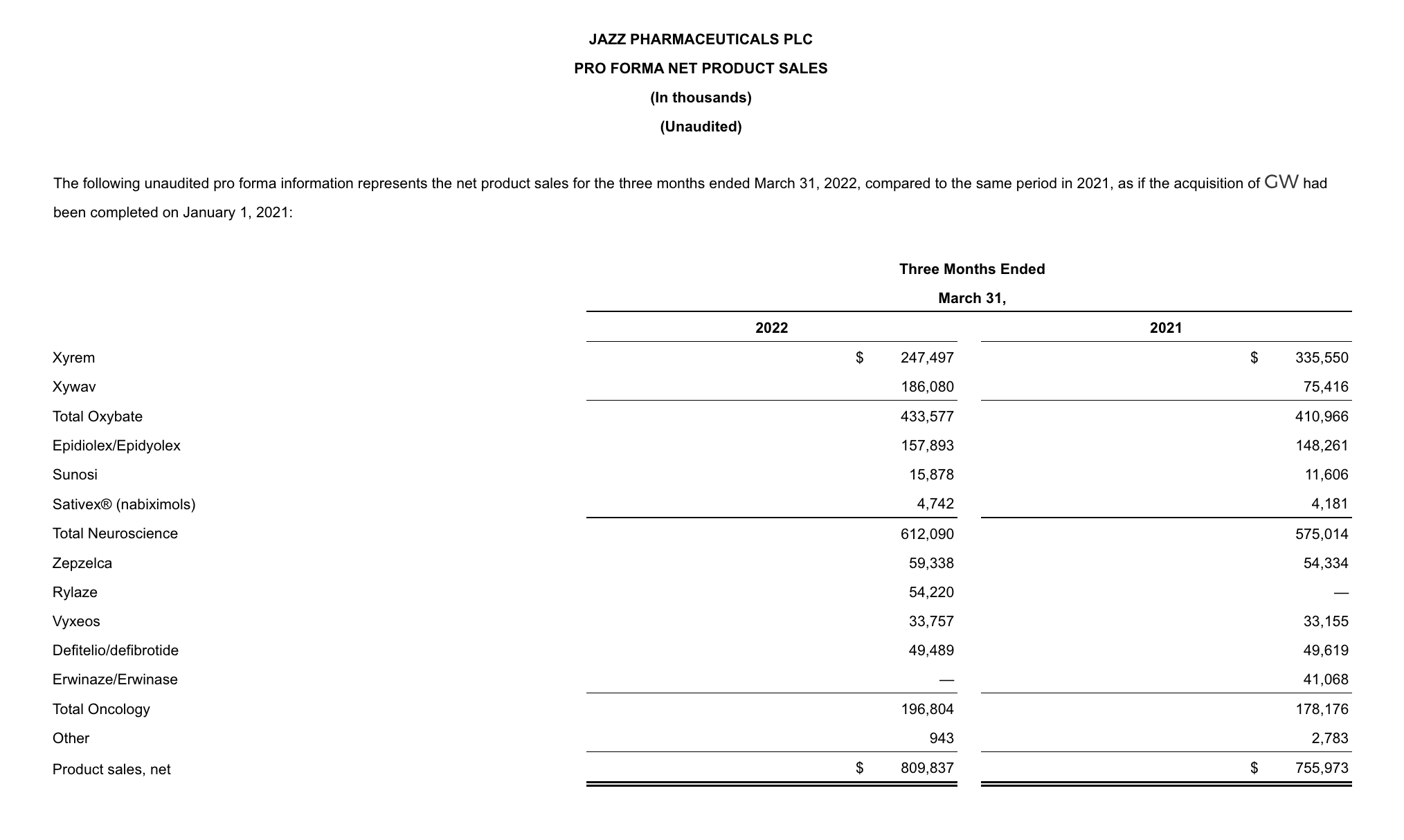
- Net product sales for the combined oxybate business increased 6% to $433.6 million in 1Q22 compared to the same period in 2021.
- Average active oxybate patients on therapy was approximately 16,650 in 1Q22, an increase of approximately 6% compared to the same period in 2021.
- Xywav net product sales increased 147% to $186.1 million in 1Q22 compared to the same period in 2021.
- There were approximately 7,800 active Xywav patients exiting 1Q22.
- Xywav has broad patent protection to 2033.
- There were approximately 7,050 narcolepsy patients taking Xywav exiting 1Q22.
- The benefits of lowering sodium intake continues to resonate with patients and prescribers. In June 2021, U.S. Food and Drug Administration (FDA) recognized seven years of Orphan Drug Exclusivity (ODE), through July 2027, for Xywav and published its summary of clinical superiority findings stating that “Xywav is clinically superior to Xyrem by means of greater safety because Xywav provides a greatly reduced chronic sodium burden compared to Xyrem.” Further, FDA stated that “the differences in the sodium content of the two products at the recommended doses will be clinically meaningful in reducing cardiovascular morbidity in a substantial proportion of patients for whom the drug is indicated.”
- Positive early launch momentum with approximately 750 IH patients taking Xywav exiting 1Q22.
- The Company launched Xywav for IH in November 2021, with initial launch efforts focused on the approximately 37,000 currently diagnosed patients in the U.S. who are actively seeking healthcare. Healthcare providers are excited to have a treatment option with positive and compelling clinical trial results that address IH and not just its symptoms.
- FDA recognized ODE for IH in January 2022, extending regulatory exclusivity to August 2028.
- Xyrem net product sales decreased 26% to $247.5 million in 1Q22 compared to the same period in 2021, reflecting the continued adoption of Xywav by patients with narcolepsy.
- Epidiolex/Epidyolex net product sales increased 6% to $157.9 million in 1Q22 compared to the same period in 2021, on a proforma basis.
- Epidiolex/Epidyolex net product sales in 4Q21 were favorably impacted by approximately $18 million, due to a temporary increase in specialty pharmacy inventory levels at the end of 2021. The majority of this increase reversed in 1Q22, reducing 1Q22 revenues.
- Excluding this impact, we saw double-digit percentage revenue growth in 1Q22 compared to 1Q21, and sequential growth in underlying demand, despite challenges posed by the Omicron variant.
- Epidyolex is now commercially available and fully reimbursed in four of the five key European markets: United Kingdom, Germany, Italy and Spain, with an anticipated launch in France in 2022. The Company has made significant progress on its European rollout with launches in Spain, Italy and Switzerland in 3Q21 and Ireland and Norway in 1Q22.
- The Company expects to initiate a Phase 3 pivotal trial of Epidiolex for Epilepsy with Myoclonic-Atonic Seizures (EMAS), the fourth target indication for Epidiolex, in 1H22.
- The Company continues to strengthen the durability of Epidiolex. Patent US 11,207,292 is Orange Book listed and extends through 2039. This patent covers the composition of the botanically derived cannabidiol (CBD) preparation used in Epidiolex and the treatment of indicated disorders using that CBD preparation.
- Zepzelca net product sales increased 9% to $59.3 million in 1Q22 compared to the same period in 2021.
- The Company is pleased to have established Zepzelca as the treatment of choice in the second-line small cell lung cancer (SCLC) setting after only eighteen months on the market.
- Zepzelca development program updates:
- In March 2022, the first patient was enrolled in the EMERGE-201 Phase 2 basket trial evaluating Zepzelca as monotherapy in select relapsed/refractory solid tumors.
- Jazz and collaborator F. Hoffmann-La Roche Ltd (Roche) have initiated a Phase 3 trial to evaluate first-line use of Zepzelca in combination with Tecentriq® (atezolizumab), compared to Tecentriq alone, as maintenance therapy in patients with extensive-stage SCLC after induction chemotherapy. The first patient was enrolled in the trial in November 2021.
- The Company’s partner, PharmaMar, initiated a confirmatory trial, LAGOON, in second-line SCLC in December 2021. If positive, this trial could confirm the benefit of Zepzelca in the treatment of SCLC when patients progress following first-line treatment with a platinum-based regimen.
- Rylaze net product sales were $54.2 million in 1Q22.
- The continued strong launch of Rylaze reflects the significant unmet patient need for a high-quality, reliable supply of Erwinia asparaginase for patients with ALL.
- In January 2022, the Company completed the submission of an sBLA to FDA seeking approval for a M/W/F IM dosing schedule for Rylaze. In April 2022, the Company also completed the submission of an sBLA to FDA seeking approval for IV administration of Rylaze. Both submissions are being reviewed under the RTOR program.
- The Company anticipates that data from the current development program will support regulatory filings in Europe in mid-2022, including IV administration, with potential for approval in 2023. The Company is also working with a partner to advance the program for potential submission, approval and launch in Japan.
- On May 4, 2022, the Company and Sumitomo Pharma Co., Ltd. announced an exclusive license agreement for DSP-0187, now called JZP441, a potent, highly selective oral orexin-2 receptor agonist designed to activate orexin signaling.
- Sumitomo Pharma initiated a Phase 1 clinical trial in Japan in November 2021 to evaluate safety, tolerability, and pharmacokinetics in healthy volunteers.
- The collaboration will leverage the Company’s substantial experience and leadership in sleep disorders to advance this therapy with the potential to improve patient care.
- Financial terms included a $50 million upfront payment to Sumitomo Pharma, and Sumitomo Pharma is eligible to receive development, regulatory and commercial milestone payments of up to $1.09 billion. Pending approval, Sumitomo Pharma is eligible to receive a tiered, low double-digit royalty on the Company’s future net sales of JZP441.
- On April 7, 2022, the Company and Werewolf Therapeutics entered into a licensing agreement under which the Company acquired exclusive global development and commercialization rights to Werewolf’s investigational molecule, WTX-613, now called JZP898, a differentiated, conditionally activated IFNα INDUKINE™ molecule.
- JZP898 is an engineered IFNα cytokine pro-drug that is activated specifically within the tumor microenvironment where it can stimulate IFNα receptors on cancer-fighting immune effector cells. The aim of JZP898 is to minimize the severe toxicities that have been observed with systemically active recombinant IFNα therapy and maximize clinical benefit when administered as monotherapy or in combination with other agents.
- Jazz expects to file an Investigational New Drug (IND) application in the U.S. in 2023.
- Financial terms included a $15 million upfront payment to Werewolf, and Werewolf is eligible to receive development, regulatory and commercial milestone payments of up to $1.26 billion. Pending approval, Werewolf is eligible to receive a tiered, mid-single-digit percentage royalty on the net sales.
- On March 28, 2022, Jazz entered into a definitive agreement to divest Sunosi to Axsome Therapeutics.
- The Company will receive an upfront payment of $53 million, a high single-digit royalty on Axsome’s U.S. net sales of Sunosi in current indications and a mid-single-digit royalty on Axsome’s U.S. net sales of Sunosi in future indications.
- The Company and Axsome are committed to ensuring that patients receive uninterrupted access to Sunosi throughout the transition.
- The companies expect the U.S. transaction to close in the second quarter of 2022 and the ex-U.S. transaction close to occur within 60 days following the close of the U.S. transaction.
- There are currently three ongoing Phase 3 trials in multiple sclerosis (MS)-related spasticity. The Company anticipates data from its first Phase 3 trial, NCT04657666, in 2Q22; supportive findings may enable a New Drug Application submission to FDA in 2022.
- Suvecaltamide, a highly selective modulator of T-type calcium channels, is in clinical development for the treatment of essential tremor.
- The Company initiated a Phase 2b trial in 4Q21 and announced that the first patient was enrolled in December 2021. Top-line data read-out is anticipated in 1H24.
- JZP150, a selective fatty acid amide hydrolase, or FAAH, inhibitor, is in clinical development for the potential treatment of post-traumatic stress disorder (PTSD).
- The Company initiated a Phase 2 trial in 4Q21 and announced that the first patient was enrolled in December 2021. Top-line data read-out is anticipated in late 2023.
- The Company received Fast Track Designation for JZP150 development in PTSD from FDA in 4Q21, underscoring the significant unmet medical needs of patients.
- JZP815 is an investigational, preclinical stage pan-RAF kinase inhibitor that targets specific components of the mitogen-activated protein kinase (MAPK) pathway, which when activated by oncogenic mutations, can be a frequent driver of human cancer.
- The pan-RAF inhibitor program is part of a novel class of next-generation precision oncology therapies that has the potential to benefit cancer patients with high unmet needs in multiple different solid tumors.
- The Company, together with our preclinical collaboration partner, Redx Pharma, presented its first preclinical data in a poster at the American Association for Cancer Research Annual Meeting in April 2022.
- JZP815 inhibited tumor growth in several RAS- and BRAF-mutated solid tumor models, and demonstrated enhanced activity when combined with other MAPK pathway inhibitors.
- The Company plans to submit an IND for JZP815 this year.
Sunosi® (solriamfetol):
- Sunosi net product sales increased by 37% to $15.9 million in 1Q22 compared to the same period in 2021.
Vyxeos® (daunorubicin and cytarabine) liposome for injection:
- Vyxeos net product sales increased 2% to $33.8 million in 1Q22 compared to the same period in 2021.
Defitelio® (defibrotide sodium) / defibrotide:
- Defitelio/defibrotide net product sales of $49.5 million in 1Q22 were consistent with the same period in 2021.
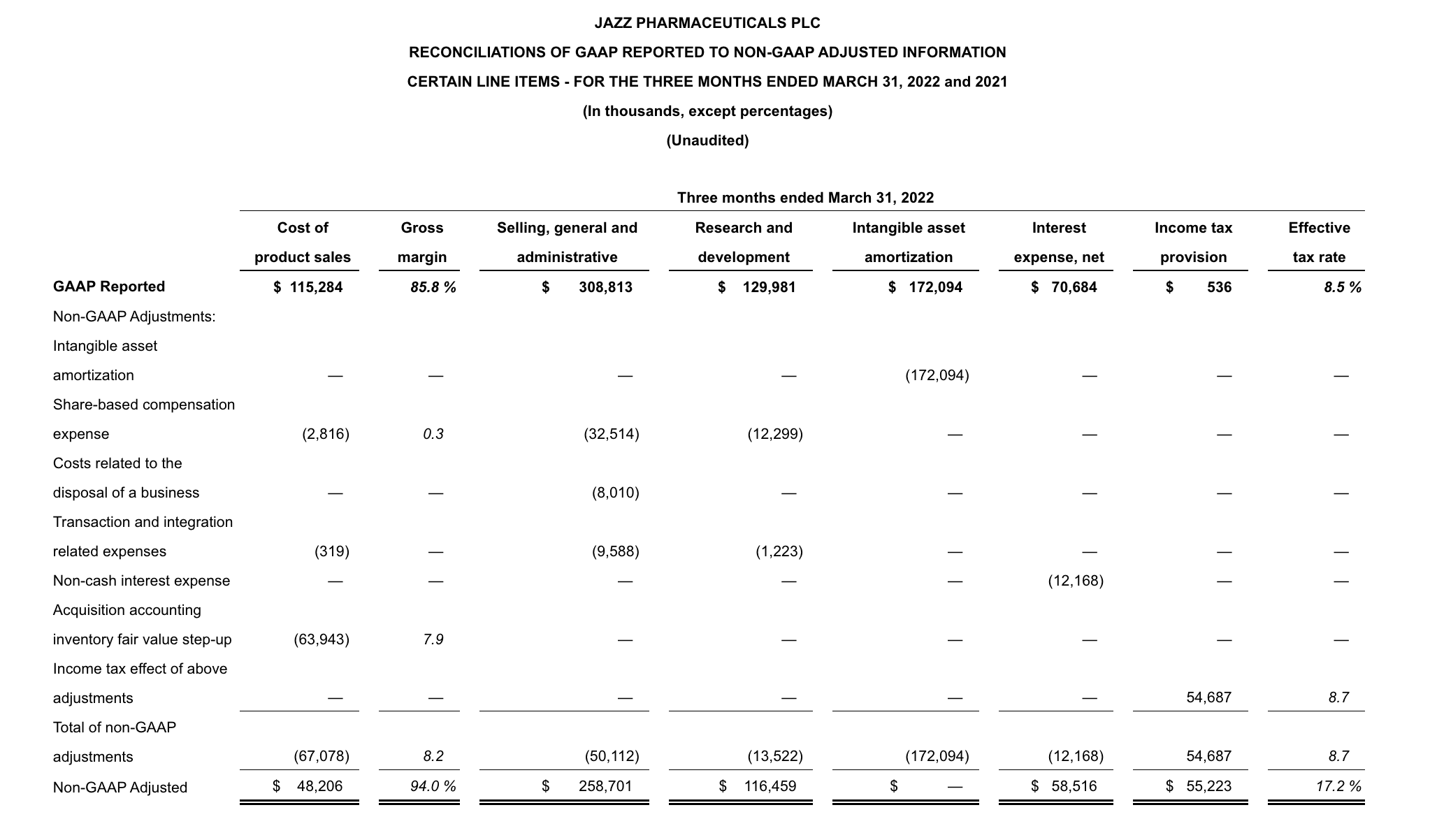
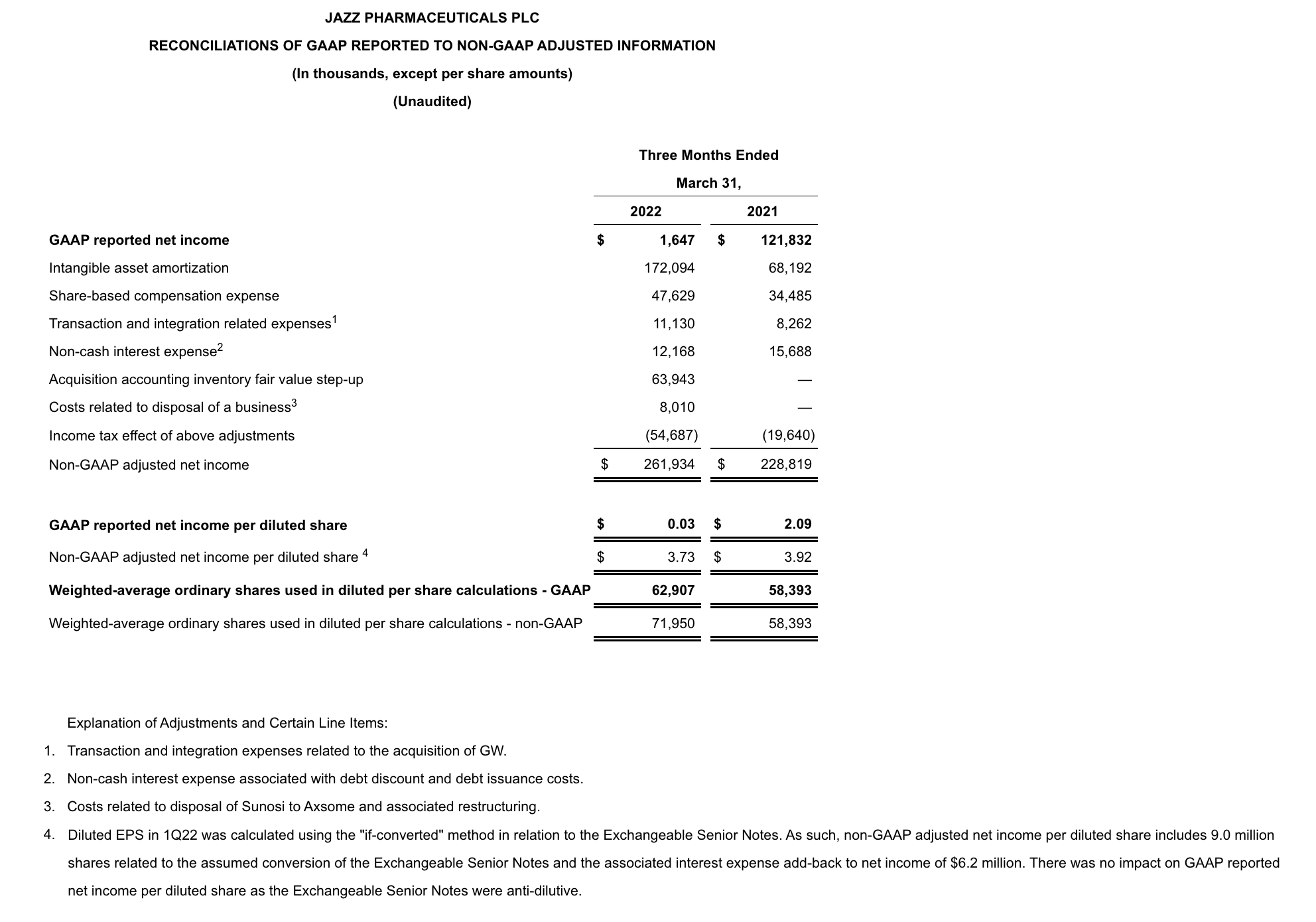
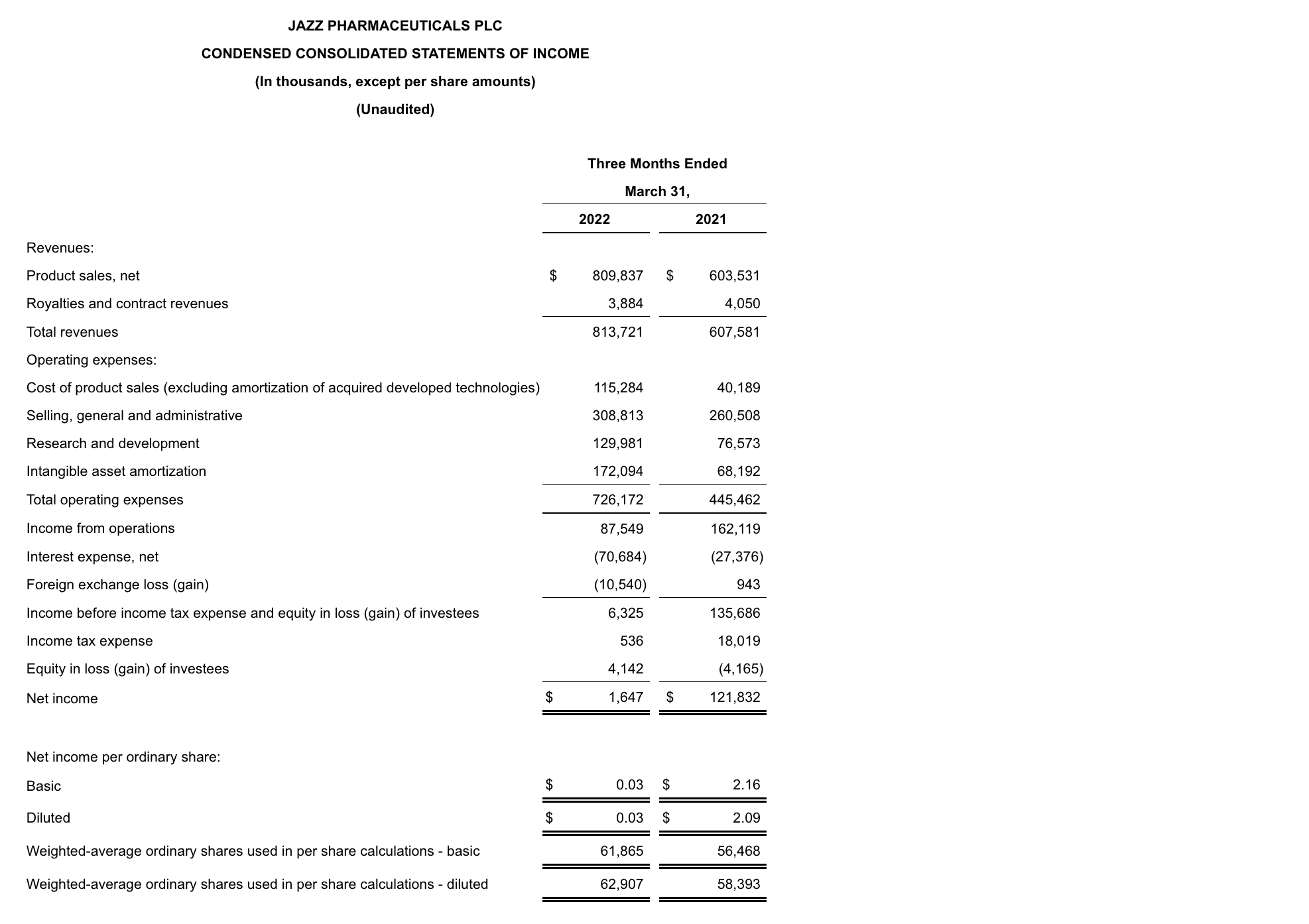
GAAP net income in 1Q22 was $1.6 million, or $0.03 per diluted share, compared to $121.8 million, or $2.09 per diluted share, for 1Q22. Non-GAAP adjusted net income in 1Q22 was $261.9 million, or $3.73 per diluted share, compared to $228.8 million, or $3.92 per diluted share, for 1Q22. Reconciliations of applicable GAAP reported to non-GAAP adjusted information are included at the end of this press release.
Total revenues increased 34% in 1Q22 compared to the same period in 2021.
- Neuroscience net product sales in 1Q22 increased 45% to $612.1 million compared to the same period in 2021 primarily driven by Epidiolex/Epidyolex net product sales in the first quarter of 2022 of $157.9 million following the acquisition of GW. In 1Q22, oxybate net product sales increased 6% to $433.6 million.
- Oncology net product sales in 1Q22 increased 10% to $196.8 million compared to the same period in 2021 primarily driven by Rylaze net product sales in 1Q22 of $54.2 million following product launch in July 2021, partially offset by Erwinaze/Erwinase net product sales in 1Q21 of $41.1 million.
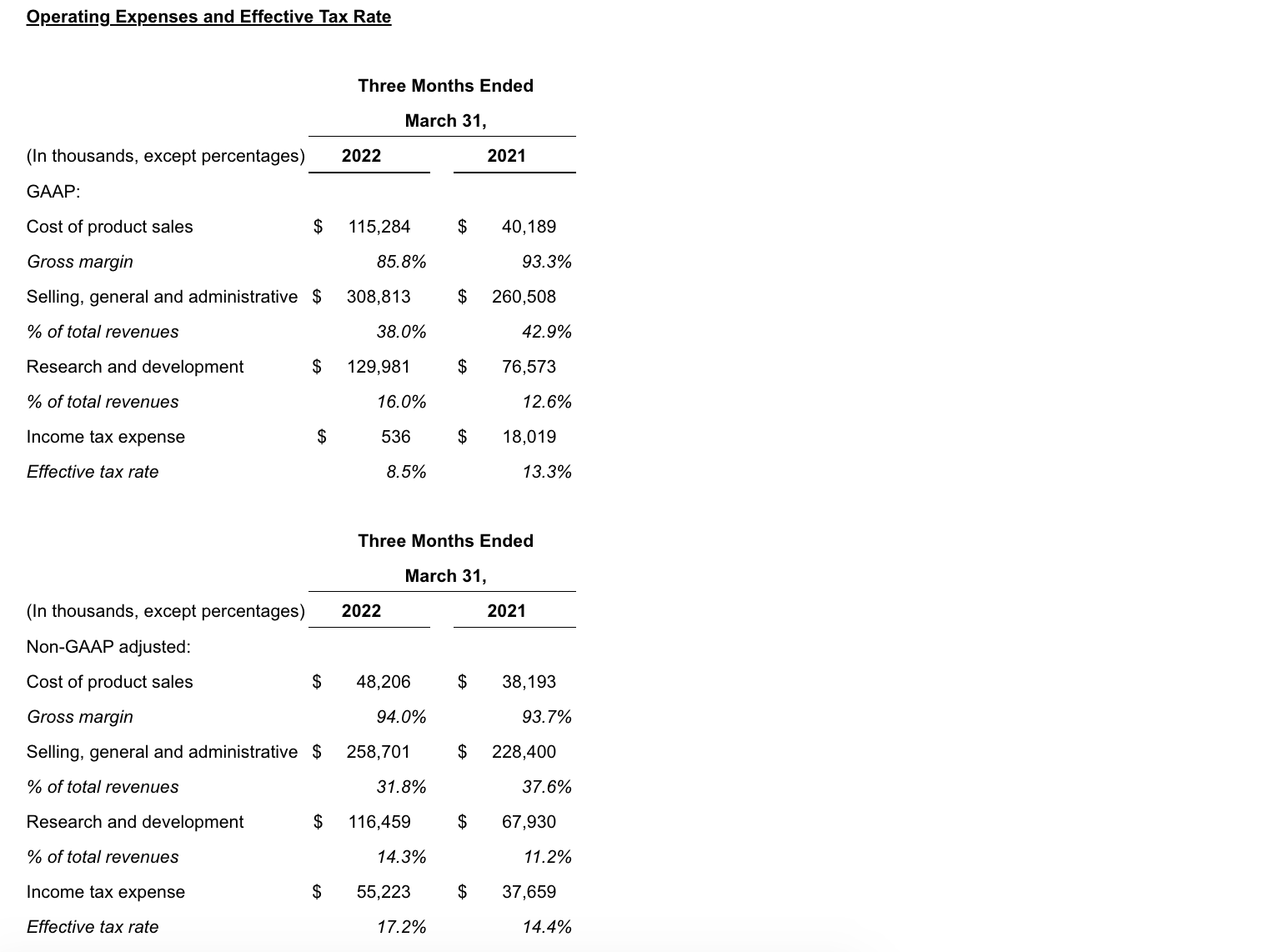
Operating expenses increased over the prior year period primarily due to the following:
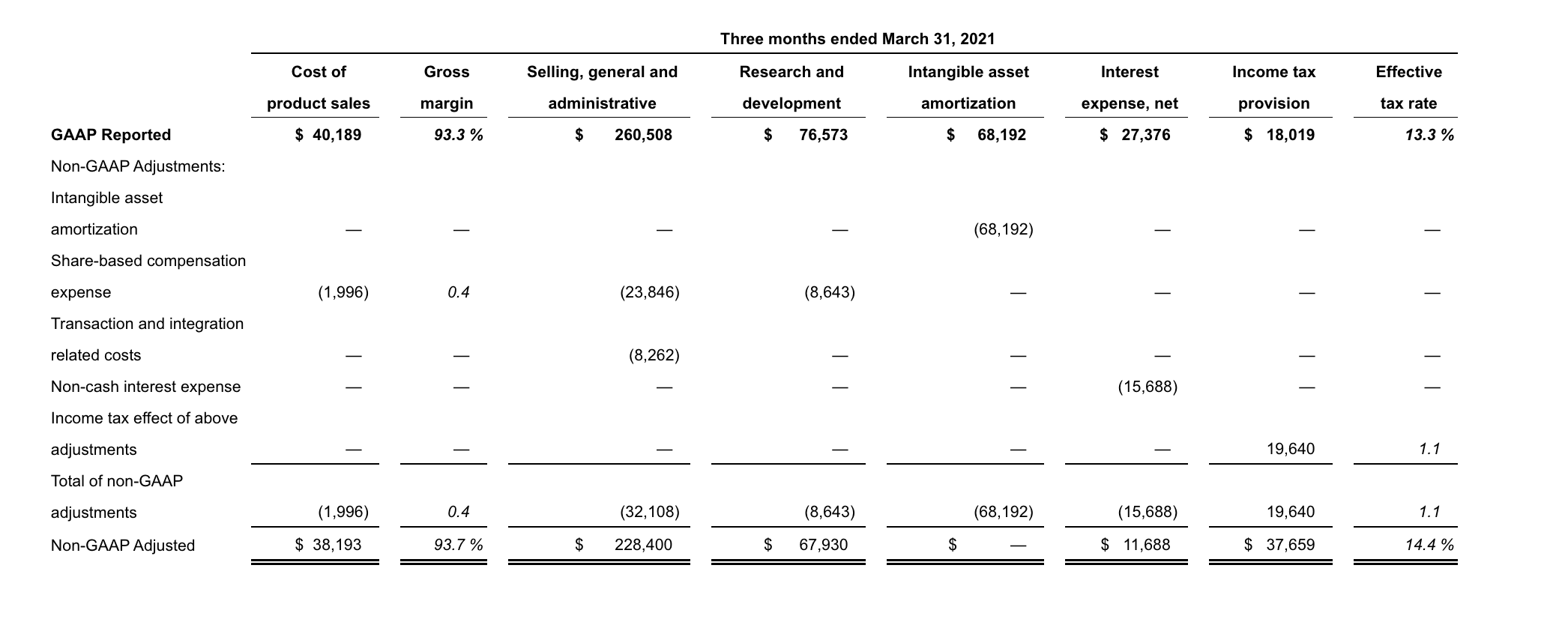
- Cost of product sales increased in 1Q22 compared to the same period in 2021, on a GAAP and on a non-GAAP adjusted basis, due to increased net product sales as a result of the acquisition of GW. In addition, acquisition accounting inventory fair value step-up expense of $63.9 million in 1Q22 impacted GAAP cost of product sales.
- Selling, general and administrative (SG&A) expenses increased in 1Q22 compared to the same period in 2021, on a GAAP and on a non-GAAP adjusted basis, primarily due to an increase in compensation-related expenses driven by higher headcount as a result of the acquisition of GW.
- Research and development (R&D) expenses increased in 1Q22 compared to the same period in 2021, on a GAAP and on a non-GAAP adjusted basis, primarily due to the addition of costs related to clinical programs for Epidiolex, nabiximols and cannabinoids, an increase in costs related to JZP150 and suvecaltamide (JZP385) and an increase in compensation-related expenses due to higher headcount primarily driven by the acquisition of GW.
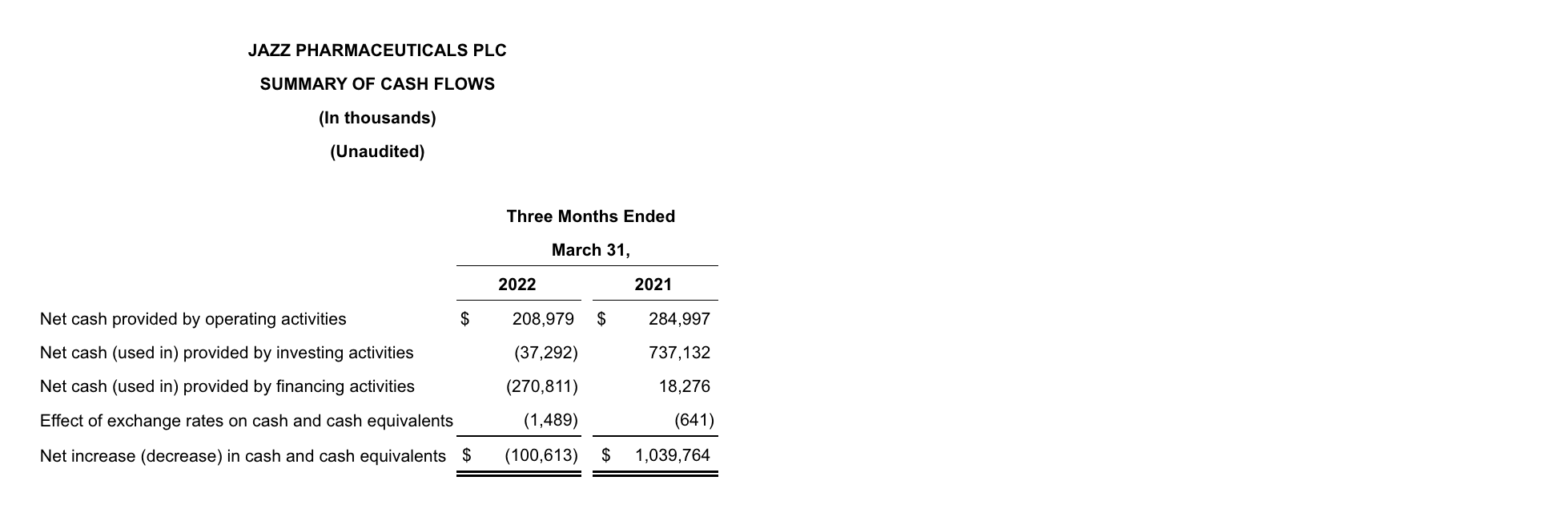
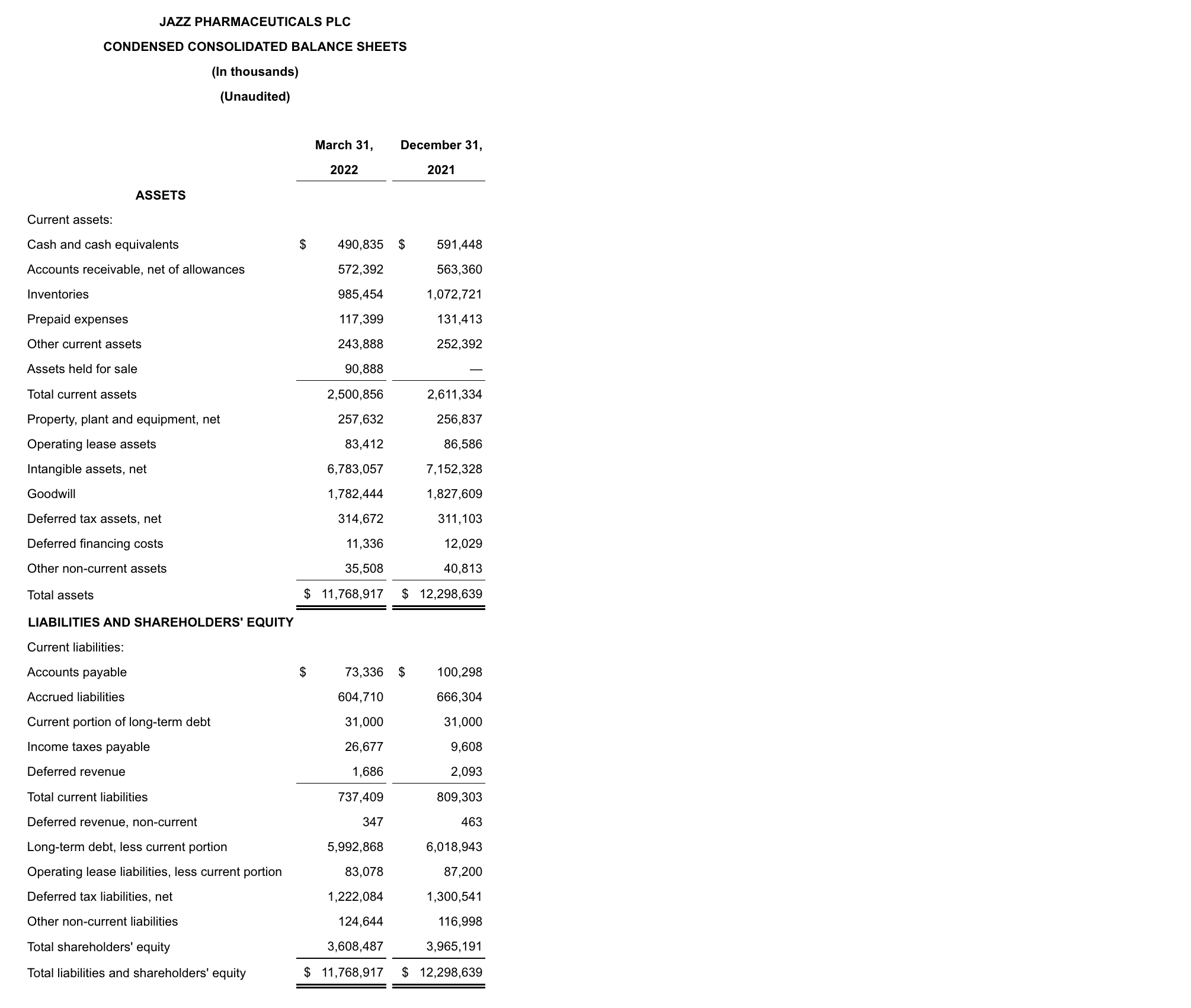
As of March 31, 2022, cash and cash equivalents were $490.8 million, and the outstanding principal balance of the Company’s long-term debt was $6.2 billion compared to $6.4 billion as of December 31, 2021. In addition, the Company had undrawn borrowing capacity under a revolving credit facility of $500.0 million. For the three months ended March 31, 2022, the Company generated $209.0 million of cash from operations. In 1Q22 the Company repaid in full the $251.0 million remaining aggregate principal amount of the Euro Term Loan B.
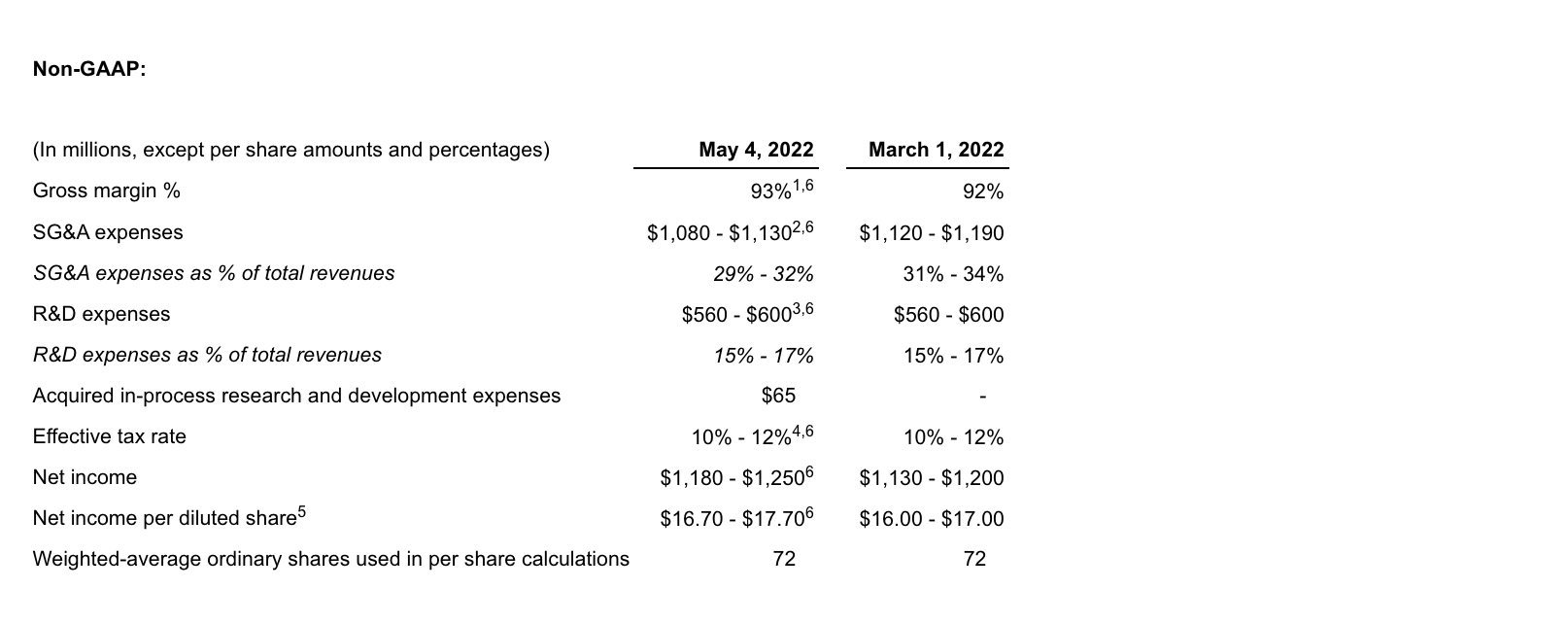 1. Excludes $305-$340 million of amortization of acquisition-related inventory fair value step-up, $13-$15 million of share-based compensation expense and $2 million of transaction and integration related expenses relating to the acquisition of GW from estimated GAAP gross margin.
1. Excludes $305-$340 million of amortization of acquisition-related inventory fair value step-up, $13-$15 million of share-based compensation expense and $2 million of transaction and integration related expenses relating to the acquisition of GW from estimated GAAP gross margin.
2. Excludes $148-$168 million of share-based compensation expense and $31-$41 million of transaction and integration related expenses relating to the acquisition of GW and $40-$50 million of costs related to the disposal of a business from estimated GAAP SG&A expenses.
3. Excludes $59-$67 million of share-based compensation expense and $2 million of transaction and integration related expenses relating to the acquisition of GW from estimated GAAP R&D expenses.
4. Excludes the income tax effect of adjustments between GAAP net income and non-GAAP adjusted net income.
5. Non-GAAP adjusted EPS guidance for 2022 reflects dilution of $2.05, at the midpoint, post adoption of ASU 2020-06. Diluted EPS calculations for 2022 include 9 million shares related to the assumed conversion of the Exchangeable Senior Notes and the associated interest expense add-back to net income of $29 million, on a GAAP basis, when dilutive, and $25 million on a non-GAAP basis, under the “if converted” method.
6. See “Non-GAAP Financial Measures” below. Reconciliations of non-GAAP adjusted guidance measures are included above and in the table titled “Reconciliation of GAAP to non-GAAP Adjusted 2022 Net Income Guidance” at the end of this press release.
Jazz Pharmaceuticals will host an investor conference call and live audio webcast today at 4:30 p.m. ET (9:30 p.m. IST) to provide a business and financial update and discuss its 2022 first quarter results. The live webcast may be accessed from the Investors section of the Company’s website at www.jazzpharmaceuticals.com. Please connect to the website prior to the start of the conference call to ensure adequate time for any software downloads that may be necessary. Investors may participate in the conference call by dialing +1 855 353 7924 in the U.S., or +1 503 343 6056 outside the U.S., and entering passcode 7492554.
A replay of the conference call will be available through May 11, 2022 by dialing +1 855 859 2056 in the U.S., or +1 404 537 3406 outside the U.S., and entering passcode 7492554. An archived version of the webcast will be available for at least one week in the Investors section of the Company’s website at www.jazzpharmaceuticals.com.
Jazz Pharmaceuticals plc (NASDAQ: JAZZ) is a global biopharmaceutical company whose purpose is to innovate to transform the lives of patients and their families. We are dedicated to developing life-changing medicines for people with serious diseases – often with limited or no therapeutic options. We have a diverse portfolio of marketed medicines and novel product candidates, from early- to late-stage development, in neuroscience and oncology. Within these therapeutic areas, we are identifying new options for patients by actively exploring small molecules and biologics, and through innovative delivery technologies and cannabinoid science. Jazz is headquartered in Dublin, Ireland and has employees around the globe, serving patients in nearly 75 countries. For more information, please visit www.jazzpharmaceuticals.com and follow @JazzPharma on Twitter.
To supplement Jazz Pharmaceuticals’ financial results and guidance presented in accordance with U.S. generally accepted accounting principles (GAAP), the Company uses certain non-GAAP (also referred to as adjusted or non-GAAP adjusted) financial measures in this press release and the accompanying tables. In particular, the Company presents non-GAAP adjusted net income (and the related per share measure) and its line item components, as well as certain non-GAAP adjusted financial measures derived therefrom, including non-GAAP adjusted gross margin percentage and non-GAAP adjusted effective tax rate. Non-GAAP adjusted net income (and the related per share measure) and its line item components exclude from GAAP reported net income (and the related per share measure) and its line item components certain items, as detailed in the reconciliation tables that follow, and in the case of non-GAAP adjusted net income (and the related per share measure), adjust for the income tax effect of the non-GAAP adjustments. In this regard, the components of non-GAAP adjusted net income, including non-GAAP adjusted cost of product sales, SG&A expenses and R&D expenses, are income statement line items prepared on the same basis as, and therefore components of, the overall non-GAAP adjusted net income measure. The Company also uses a pro forma non-GAAP net leverage ratio calculated as net debt (defined as total GAAP debt net of cash and cash equivalents) divided by Adjusted EBITDA for the most recent period of four consecutive completed fiscal quarters. EBITDA is defined as net income (loss) before income taxes, interest expense, depreciation and amortization. Adjusted EBITDA is defined as EBITDA further adjusted to exclude certain other charges and adjustments as detailed in the pro forma non-GAAP net leverage ratio reconciliation table that follows, and is calculated in accordance with the definition of Adjusted Consolidated EBITDA as set out in the Company’s credit agreement entered into in May 2021 (the Credit Agreement). Investors should note that reconciliations of certain forward-looking or projected non-GAAP financial measures to their most comparable GAAP financial measures cannot be provided because the Company cannot do so without unreasonable efforts due to the unavailability of information needed to calculate reconciling items and due to the variability, complexity and limited visibility of comparable GAAP measures and the reconciling items that would be excluded from the non-GAAP financial measures in the future. Specifically, reconciliations of the components of projected pro forma non-GAAP net leverage ratio to their most comparable GAAP financial measures is not provided because the quantification of projected GAAP total debt and the reconciling items between projected non-GAAP net adjusted debt and projected GAAP total debt cannot be reasonably calculated or predicted at this time without unreasonable efforts. Such unavailable information could be significant such that actual GAAP total debt net of cash and cash equivalents would vary significantly from projected non-GAAP net adjusted debt used to calculate projected pro forma non-GAAP net leverage ratio.
The Company believes that each of these non-GAAP financial measures provides useful supplementary information to, and facilitates additional analysis by, investors and analysts and that each of these non-GAAP financial measures, when considered together with the Company’s financial information prepared in accordance with GAAP, can enhance investors’ and analysts’ ability to meaningfully compare the Company’s results from period to period and to its forward-looking guidance, to identify operating trends in the Company’s business and to understand the Company’s ability to delever. In addition, these non-GAAP financial measures are regularly used by investors and analysts to model and track the Company’s financial performance. Jazz Pharmaceuticals’ management also regularly uses these non-GAAP financial measures internally to understand, manage and evaluate the Company’s business and to make operating decisions, and compensation of executives is based in part on certain of these non-GAAP financial measures. Because these non-GAAP financial measures are important internal measurements for Jazz Pharmaceuticals’ management, the Company also believes that these non-GAAP financial measures are useful to investors and analysts since these measures allow for greater transparency with respect to key financial metrics the Company uses in assessing its own operating performance and making operating decisions. These non-GAAP financial measures are not meant to be considered in isolation or as a substitute for comparable GAAP measures; should be read in conjunction with the Company’s consolidated financial statements prepared in accordance with GAAP; have no standardized meaning prescribed by GAAP; and are not prepared under any comprehensive set of accounting rules or principles in the reconciliation tables that follow. In addition, from time to time in the future there may be other items that the Company may exclude for purposes of its non-GAAP financial measures; and the Company has ceased, and may in the future cease, to exclude items that it has historically excluded for purposes of its non-GAAP financial measures. Likewise, the Company may determine to modify the nature of its adjustments to arrive at its non-GAAP financial measures. Because of the non-standardized definitions of non-GAAP financial measures, the non-GAAP financial measures as used by Jazz Pharmaceuticals in this press release and the accompanying tables have limits in their usefulness to investors and may be calculated differently from, and therefore may not be directly comparable to, similarly titled measures used by other companies.
This press release contains forward-looking statements, including, but not limited to, statements related to: the Company’s growth prospects and future financial and operating results, including the Company’s 2022 financial guidance and the Company’s expectations related thereto; the proposed divestiture of Sunosi to Axsome, the anticipated upfront payment and royalties to be received by Jazz in connection therewith and the other anticipated benefits thereof; statements related to DSP-0187’s potential application for the treatment of sleep disorders; the potential successful future development, manufacturing, regulatory and commercialization activities; potential future payments by Jazz Pharmaceuticals to Sumitomo Pharma and Werewolf for development, regulatory and commercial milestones as well as tiered royalties based on future net sales; statements related to WTX-613’s demonstrated anti-tumor activity; the Company’s expectation of sustainable growth and enhanced value as part of its Vision 2025; growing and diversifying the Company’s revenue, investing in its pipeline of novel therapies, and delivering innovative therapies for patients; the Company’s ability to realize the commercial potential of its products, including the blockbuster potential of Epidiolex; the Company’s views and expectations relating to its patent portfolio, including with respect to expected patent protection; planned or anticipated clinical trial events, including with respect to initiations, enrollment and data read-outs, and the anticipated timing thereof; the Company’s clinical trials confirming clinical benefit or enabling regulatory submissions; planned or anticipated regulatory submissions and filings, including for nabiximols and Rylaze, and the anticipated timing thereof; potential regulatory approvals, including for Rylaze; the anticipated launch of Epidyolex in France in 2022; and other statements that are not historical facts. These forward-looking statements are based on the Company’s current plans, objectives, estimates, expectations and intentions and inherently involve significant risks and uncertainties.
Actual results and the timing of events could differ materially from those anticipated in such forward- looking statements as a result of these risks and uncertainties, which include, without limitation, risks and uncertainties associated with: Jazz’s and Axsome’s ability to complete the proposed divestiture of Sunosi on the proposed terms or on the anticipated timeline, or at all, including risks and uncertainties related to the expiration or securing early termination of the applicable waiting period under the HSR act; maintaining or increasing sales of and revenue from the Company’s oxybate products, Zepzelca and other key marketed products; effectively launching and commercializing the Company’s other products and product candidates; obtaining and maintaining adequate coverage and reimbursement for the Company’s products; the time-consuming and uncertain regulatory approval process, including the risk that the Company’s current and/or planned regulatory submissions may not be submitted, accepted or approved by applicable regulatory authorities in a timely manner or at all, including the risk that the Company’s sBLA seeking approval for a revised dosing label for Rylaze may not be approved by FDA in a timely manner or at all; the costly and time-consuming pharmaceutical product development and the uncertainty of clinical success, including risks related to failure or delays in successfully initiating or completing clinical trials and assessing patients such as those being experienced, and expected to continue to be experienced, by the Company as a result of the effects of the COVID-19 pandemic; the Company’s failure to realize the expected benefits of its acquisition of GW Pharmaceuticals, including the failure to realize the blockbuster potential of Epidiolex and the risk that the legacy GW Pharmaceuticals business will not be integrated successfully or that such integration may be more difficult, time-consuming or costly than expected; the ultimate duration and severity of the COVID-19 pandemic and resulting global economic, financial, and healthcare system disruptions and the current and potential future negative impacts to the Company’s business operations and financial results; regulatory initiatives and changes in tax laws; market volatility; protecting and enhancing the Company’s intellectual property rights and the Company’s commercial success being dependent upon the Company obtaining, maintaining and defending intellectual property protection for its products and product candidates; delays or problems in the supply or manufacture of the Company’s products and product candidates; complying with applicable U.S. and non-U.S. regulatory requirements, including those governing the research, development, manufacturing and distribution of controlled substances; government investigations, legal proceedings and other actions; identifying and acquiring, in-licensing or developing additional products or product candidates, financing these transactions and successfully integrating acquired product candidates, products and businesses; the Company’s ability to realize the anticipated benefits of its collaborations and license agreements with third parties; the sufficiency of the Company’s cash flows and capital resources to fund its debt service obligations, de-lever and meet its stated leverage targets; the Company’s ability to achieve expected future financial performance and results and the uncertainty of future tax, accounting and other provisions and estimates; the possibility that, if the Company does not achieve the perceived benefits of the acquisition of GW Pharmaceuticals as rapidly or to the extent anticipated by financial analysts or investors, the market price of the Company’s ordinary shares could decline; the Company’s ability to achieve expected future financial performance and results and the uncertainty of future tax and other provisions and estimates; the Company’s ability to meet its projected long-term goals and objectives, including as part of Vision 2025, in the time periods that the Company anticipates, or at all, and the inherent uncertainty and significant judgments and assumptions underlying the Company’s long-term goals and objectives; and other risks and uncertainties affecting the Company, including those described from time to time under the caption “Risk Factors” and elsewhere in Jazz Pharmaceuticals’ Securities and Exchange Commission filings and reports, including the Company’s Annual Report on Form 10-K for the year ended December 31, 2021, and future filings and reports by the Company. Other risks and uncertainties of which the Company is not currently aware may also affect the Company’s forward-looking statements and may cause actual results and the timing of events to differ materially from those anticipated.
Contacts:
Investors:
Andrea N. Flynn, Ph.D.
Vice President, Head, Investor Relations
Jazz Pharmaceuticals plc
InvestorInfo@jazzpharma.com
Ireland +353 1 634 3211
U.S. +1 650 496 2717
Media:
Kristin Bhavnani
Head of Global Corporate Communications
Jazz Pharmaceuticals plc
CorporateAffairsMediaInfo@jazzpharma.com
Ireland +353 1 637 2141
U.S. +1 215 867 4948
SOURCE Jazz Pharmaceuticals plc