 Organigram Reports Q3 Financial Results – Increased Product Quality – 25% Patient Growth
Organigram Reports Q3 Financial Results – Increased Product Quality – 25% Patient Growth
MONCTON, NEW BRUNSWICK–(Marketwired – July 28, 2017) – Organigram Holdings Inc. (TSX VENTURE:OGI)(OTCQB:OGRMF) (“Organigram” or the “Company”) announces the financial results for the quarter ending May 31, 2017.
Organigram’s financial results for the quarter completed May 31, 2017 included expenses associated with the implementation of an industry leading quality assurance program (the “QA Program”). The QA Program was implemented to ensure that the Company produces the highest quality product available in the marketplace and in response to a previously announced voluntary recall of certain product produced in 2016.
As a result of Organigram’s increased focus on product quality and safety with the stated objective of establishing a leadership position in such regard within the industry, the Company focused on introducing product of the highest quality to the market. In practice, the program effectively identified product lots that were to be removed from inventory well before being introduced to the marketplace. The successful implementation of this program resulted in a reduction in inventory value for the quarter.
Organigram is pleased with the results and progress made during the quarter from an operational perspective and management is confident that these actions build the foundation for long-term success for the Company. This focus on product quality contributed to a 25% net increase in registered patients in Q3 when compared to Q2.
Additionally, the Company continued to make significant headway in increasing its overall production capacity and ensuring that it is prepared to be a market leader in the proposed adult-use recreational market in anticipation of a late-June 2018 adult recreational program launch date.
Selected highlights from the quarter include the following:
Our number one priority in Q3 was ensuring that product quality and safety remain a stated objective of Organigram. Through the implementation of a tier-one quality assurance program and improved production techniques, the Company will ensure that only the finest quality product is sold to clients of Organigram.
 Greg Engel, Chief Executive Officer of Organigram
Greg Engel, Chief Executive Officer of Organigram
We are confident that these actions result in Organigram being well poised for future success, our facility is among the best across Canada and with our geographical-based cost advantages and improved product quality, I am very optimistic about the ability of the Company to maximize stakeholder value going forward.
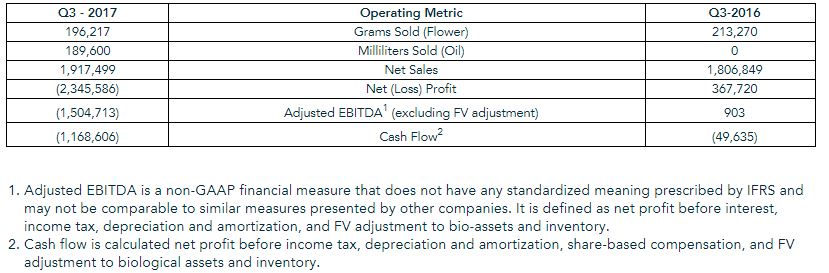
Due to the implementation of the enhanced QA Program which resulted in less product being introduced to the market than originally contemplated, Organigram recognized indirect production expenses of $1.1 million related to inventory destruction, and a loss of $1.6 million due to fair value adjustment to biological assets and net realizable reduction to inventory. As a result, the Company generated adjusted EBITDA of ($1.5) million and cash flow of ($1.1) million. Organigram’s cash and short-term investment balance was in excess of $48 million as at May 31, 2017.
Again in Q3, Organigram made significant progress with its onsite expansion initiatives as the Company has continued with progress towards increasing production capacity by an additional 10,000 kg’s annually by the end of December, 2017. Due to the anticipated delay in the timing for the allowance of edible products, the construction and implementation of the planned commercial scale oils and extracts manufacturing facility has been deferred and the recently announced high-volume supercritical CO2 equipment from Advanced Extraction Systems (“AES”) will be placed in our existing facility.
For more information, visit www.organigram.ca
About Organigram Holdings Inc.
Organigram Holdings Inc. is a TSX Venture Exchange listed company whose wholly owned subsidiary, Organigram Inc., is a licensed producer of medical marijuana in Canada. Organigram is focused on producing the highest quality, condition specific medical marijuana for patients in Canada. Organigram’s facility is located in Moncton, New Brunswick and the Company is regulated by the Access to Cannabis for Medical Purposes Regulations (“ACMPR”).
Original press release: http://www.marketwired.com/press-release/organigram-reports-q3-financial-results-increased-product-quality-25-patient-growth-tsx-venture-ogi-2228082.htm
